8-year retrospective analysis of intravenous arginine therapy for acute metabolic strokes in pediatric mitochondrial disease
- PMID: 29428506
- PMCID: PMC5849405
- DOI: 10.1016/j.ymgme.2018.01.010
8-year retrospective analysis of intravenous arginine therapy for acute metabolic strokes in pediatric mitochondrial disease
Abstract
Background: Intravenous (IV) arginine has been reported to ameliorate acute metabolic stroke symptoms in adult patients with Mitochondrial Encephalopathy with Lactic Acidosis and Stroke-like Episodes (MELAS) syndrome, where its therapeutic benefit is postulated to result from arginine acting as a nitric oxide donor to reverse vasospasm. Further, reduced plasma arginine may occur in mitochondrial disease since the biosynthesis of arginine's precursor, citrulline, requires ATP. Metabolic strokes occur across a wide array of primary mitochondrial diseases having diverse molecular etiologies that are likely to share similar pathophysiologic mechanisms. Therefore, IV arginine has been increasingly used for the acute clinical treatment of metabolic stroke across a broad mitochondrial disease population.
Methods: We performed retrospective analysis of a large cohort of subjects who were under 18 years of age at IRB #08-6177 study enrollment and had molecularly-confirmed primary mitochondrial disease (n = 71, excluding the common MELAS m.3243A>G mutation). 9 unrelated subjects in this cohort received acute arginine IV treatment for one or more stroke-like episodes (n = 17 total episodes) between 2009 and 2016 at the Children's Hospital of Philadelphia. Retrospectively reviewed data included subject genotype, clinical symptoms, age, arginine dosing, neuroimaging (if performed), prophylactic therapies, and adverse events.
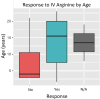
Results: Genetic etiologies of subjects who presented with acute metabolic strokes included 4 mitochondrial DNA (mtDNA) pathogenic point mutations, 1 mtDNA deletion, and 4 nuclear gene disorders. Subject age ranged from 19 months to 23 years at the time of any metabolic stroke episode (median, 8 years). 3 subjects had recurrent stroke episodes. 70% of subjects were on prophylactic arginine or citrulline therapy at the time of a stroke-like episode. IV arginine was initiated on initial presentation in 65% of cases. IV arginine was given for 1-7 days (median, 1 day). A positive clinical response to IV arginine occurred in 47% of stroke-like episodes; an additional 6% of episodes showed clinical benefit from multiple simultaneous treatments that included arginine, confounding sole interpretation of arginine effect. All IV arginine-responsive stroke-like episodes (n = 8) received treatment immediately on presentation (p = .003). Interestingly, the presence of unilateral symptoms strongly predicted arginine response (p = .02, Chi-Square); however, almost all of these cases immediately received IV arginine, confounding interpretation of causality direction. Suggestive trends toward increased IV arginine response were seen in subjects with mtDNA relative to nDNA mutations and in older pediatric subjects, although statistical significance was not reached possibly due to small sample size. No adverse events, including hypotensive episodes, from IV arginine therapy were reported.
Conclusions: Single-center retrospective analysis suggests that IV arginine therapy yields significant therapeutic benefit with little risk in pediatric mitochondrial disease stroke subjects across a wide range of genetic etiologies beyond classical MELAS. Acute hemiplegic stroke, in particular, was highly responsive to IV arginine treatment. Prospective studies with consistent arginine dosing, and pre- and post-neuroimaging, will further inform the clinical utility of IV arginine therapy for acute metabolic stroke in pediatric mitochondrial disease.
Keywords: Brain disease; Inborn error; Leigh syndrome; Metabolic stroke; Mitochondrial encephalomyopathies; Treatment.
Copyright © 2018 Elsevier Inc. All rights reserved.
Figures


References
-
- Morin C, Dubé J, Robinson BH, Lacroix J, Michaud J, De Braekeleer M, Geoffroy G, Lortie A, Blanchette C, Lambert MA, Mitchell GA. Stroke-like episodes in autosomal recessive cytochrome oxidase deficiency. Ann Neurol. 1999;45:389–92. http://www.ncbi.nlm.nih.gov/pubmed/10072055 (accessed May 28, 2017) - PubMed
-
- Furuya H, Sugimura T, Yamada T, Hayashi K, Kobayashi T. A case of incomplete Kearns-Sayre syndrome with a stroke like episode. Rinsho Shinkeigaku. 1997;37:680–4. http://www.ncbi.nlm.nih.gov/pubmed/9404143 (accessed May 28, 2017) - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

