4-PBA ameliorates cellular homeostasis in fibroblasts from osteogenesis imperfecta patients by enhancing autophagy and stimulating protein secretion
- PMID: 29432813
- PMCID: PMC5908783
- DOI: 10.1016/j.bbadis.2018.02.002
4-PBA ameliorates cellular homeostasis in fibroblasts from osteogenesis imperfecta patients by enhancing autophagy and stimulating protein secretion
Abstract
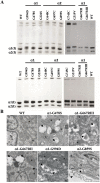
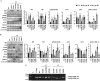
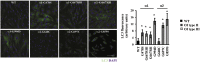
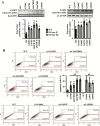
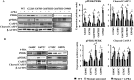
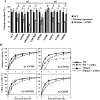
The clinical phenotype in osteogenesis imperfecta (OI) is attributed to the dominant negative function of mutant type I collagen molecules in the extracellular matrix, by altering its structure and function. Intracellular retention of mutant collagen has also been reported, but its effect on cellular homeostasis is less characterized. Using OI patient fibroblasts carrying mutations in the α1(I) and α2(I) chains we demonstrate that retained collagen molecules are responsible for endoplasmic reticulum (ER) enlargement and activation of the unfolded protein response (UPR) mainly through the eukaryotic translation initiation factor 2 alpha kinase 3 (PERK) branch. Cells carrying α1(I) mutations upregulate autophagy, while cells with α2(I) mutations only occasionally activate the autodegradative response. Despite the autophagy activation to face stress conditions, apoptosis occurs in all mutant fibroblasts. To reduce cellular stress, mutant fibroblasts were treated with the FDA-approved chemical chaperone 4-phenylbutyric acid. The drug rescues cell death by modulating UPR activation thanks to both its chaperone and histone deacetylase inhibitor abilities. As chaperone it increases general cellular protein secretion in all patients' cells as well as collagen secretion in cells with the most C-terminal mutation. As histone deacetylase inhibitor it enhances the expression of the autophagic gene Atg5 with a consequent stimulation of autophagy. These results demonstrate that the cellular response to ER stress can be a relevant target to ameliorate OI cell homeostasis.
Keywords: Autophagy; Chemical chaperone; Collagen; Endoplasmic reticulum stress; Osteogenesis imperfecta; Unfolded protein response.
Copyright © 2018 The Authors. Published by Elsevier B.V. All rights reserved.
Figures


Similar articles
-
4-Phenylbutyric acid enhances the mineralization of osteogenesis imperfecta iPSC-derived osteoblasts.J Biol Chem. 2021 Jan-Jun;296:100027. doi: 10.1074/jbc.RA120.014709. Epub 2020 Nov 23. J Biol Chem. 2021. PMID: 33154166 Free PMC article.
-
Cellular stress due to impairment of collagen prolyl hydroxylation complex is rescued by the chaperone 4-phenylbutyrate.Dis Model Mech. 2019 Jun 20;12(6):dmm038521. doi: 10.1242/dmm.038521. Dis Model Mech. 2019. PMID: 31171565 Free PMC article.
-
A novel chemical chaperone ameliorates osteoblast homeostasis and extracellular matrix in osteogenesis imperfecta.Life Sci. 2025 Jan 15;361:123320. doi: 10.1016/j.lfs.2024.123320. Epub 2024 Dec 18. Life Sci. 2025. PMID: 39706289
-
Osteogenesis imperfecta: recent findings shed new light on this once well-understood condition.Genet Med. 2009 Jun;11(6):375-85. doi: 10.1097/GIM.0b013e3181a1ff7b. Genet Med. 2009. PMID: 19533842 Review.
-
The therapeutic effects of 4-phenylbutyric acid in maintaining proteostasis.Int J Biochem Cell Biol. 2015 Apr;61:45-52. doi: 10.1016/j.biocel.2015.01.015. Epub 2015 Feb 7. Int J Biochem Cell Biol. 2015. PMID: 25660369 Review.
Cited by
-
4-Phenylbutyric acid enhances the mineralization of osteogenesis imperfecta iPSC-derived osteoblasts.J Biol Chem. 2021 Jan-Jun;296:100027. doi: 10.1074/jbc.RA120.014709. Epub 2020 Nov 23. J Biol Chem. 2021. PMID: 33154166 Free PMC article.
-
Medical Management for Fracture Prevention in Children with Osteogenesis Imperfecta.Calcif Tissue Int. 2024 Dec;115(6):812-827. doi: 10.1007/s00223-024-01202-7. Epub 2024 Mar 29. Calcif Tissue Int. 2024. PMID: 38553634 Free PMC article. Review.
-
Zebrafish as a model to study autophagy and its role in skeletal development and disease.Histochem Cell Biol. 2020 Nov;154(5):549-564. doi: 10.1007/s00418-020-01917-2. Epub 2020 Sep 11. Histochem Cell Biol. 2020. PMID: 32915267 Free PMC article. Review.
-
Dissecting the phenotypic variability of osteogenesis imperfecta.Dis Model Mech. 2022 May 1;15(5):dmm049398. doi: 10.1242/dmm.049398. Epub 2022 May 16. Dis Model Mech. 2022. PMID: 35575034 Free PMC article.
-
Emerging Landscape of Osteogenesis Imperfecta Pathogenesis and Therapeutic Approaches.ACS Pharmacol Transl Sci. 2024 Jan 2;7(1):72-96. doi: 10.1021/acsptsci.3c00324. eCollection 2024 Jan 12. ACS Pharmacol Transl Sci. 2024. PMID: 38230285 Free PMC article. Review.
References
-
- Taga Y., Kusubata M., Ogawa-Goto K., Hattori S. Site-specific quantitative analysis of overglycosylation of collagen in osteogenesis imperfecta using hydrazide chemistry and SILAC. J. Proteome Res. 2013;12:2225–2232. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials

