Annual changes in rotavirus hospitalization rates before and after rotavirus vaccine implementation in the United States
- PMID: 29444124
- PMCID: PMC5812572
- DOI: 10.1371/journal.pone.0191429
Annual changes in rotavirus hospitalization rates before and after rotavirus vaccine implementation in the United States
Abstract
Background: Hospitalizations for rotavirus and acute gastroenteritis (AGE) have declined in the US with rotavirus vaccination, though biennial peaks in incidence in children aged less than 5 years occur. This pattern may be explained by lower rotavirus vaccination coverage in US children (59% to 73% from 2010-2015), resulting in accumulation of susceptible children over two successive birth cohorts.
Methods: Retrospective cohort analysis of claims data of commercially insured US children aged <5 years. Age-stratified hospitalization rates for rotavirus and for AGE from the 2002-2015 rotavirus seasons were examined. Median age and rotavirus vaccination coverage for biennial rotavirus seasons during pre-vaccine (2002-2005), early post-vaccine (2008-2011) and late post-vaccine (2012-2015) years.
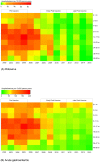
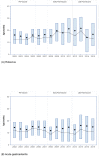
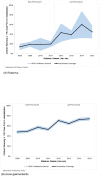
Results: Age-stratified hospitalization rates decreased from pre-vaccine to early post-vaccine and then to late post-vaccine years. The clearest biennial pattern in hospitalization rates is the early post-vaccine period, with higher rates in 2009 and 2011 than in 2008 and 2010. The pattern diminishes in the late post-vaccine period. For rotavirus hospitalizations, the median age and the difference in age between biennial seasons was highest during the early post-vaccine period; these differences were not observed for AGE hospitalizations. There was no significant difference in vaccination coverage between biennial seasons.
Conclusions: These observations provide conflicting evidence that incomplete vaccine coverage drove the biennial pattern in rotavirus hospitalizations that has emerged with rotavirus vaccination in the US. As this pattern is diminishing with higher vaccine coverage in recent years, further increases in vaccine coverage may reach a threshold that eliminates peak seasons in hospitalizations.
Conflict of interest statement
Figures
References
-
- Parashar UD, Alexander JP, Glass RI. Prevention of rotavirus gastroenteritis among infants and children. Recommendations of the Advisory Committee on Immunization Practices (ACIP). MMWR Recommendations and reports: Morbidity and mortality weekly report Recommendations and reports / Centers for Disease Control. 2006;55(Rr-12):1–13. Epub 2006/08/12. . - PubMed
-
- Shah MP, Tate JE, Steiner CA, Parashar UD. Decline in Emergency Department Visits for Acute Gastroenteritis Among Children in 10 US States After Implementation of Rotavirus Vaccination, 2003 to 2013. The Pediatric infectious disease journal. 2016;35(7):782–6. Epub 2016/04/19. 10.1097/INF.0000000000001175 . - DOI - PMC - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials

