Interacting-heads motif has been conserved as a mechanism of myosin II inhibition since before the origin of animals
- PMID: 29444861
- PMCID: PMC5834683
- DOI: 10.1073/pnas.1715247115
Interacting-heads motif has been conserved as a mechanism of myosin II inhibition since before the origin of animals
Abstract
Electron microscope studies have shown that the switched-off state of myosin II in muscle involves intramolecular interaction between the two heads of myosin and between one head and the tail. The interaction, seen in both myosin filaments and isolated molecules, inhibits activity by blocking actin-binding and ATPase sites on myosin. This interacting-heads motif is highly conserved, occurring in invertebrates and vertebrates, in striated, smooth, and nonmuscle myosin IIs, and in myosins regulated by both Ca2+ binding and regulatory light-chain phosphorylation. Our goal was to determine how early this motif arose by studying the structure of inhibited myosin II molecules from primitive animals and from earlier, unicellular species that predate animals. Myosin II from Cnidaria (sea anemones, jellyfish), the most primitive animals with muscles, and Porifera (sponges), the most primitive of all animals (lacking muscle tissue) showed the same interacting-heads structure as myosins from higher animals, confirming the early origin of the motif. The social amoeba Dictyostelium discoideum showed a similar, but modified, version of the motif, while the amoeba Acanthamoeba castellanii and fission yeast (Schizosaccharomyces pombe) showed no head-head interaction, consistent with the different sequences and regulatory mechanisms of these myosins compared with animal myosin IIs. Our results suggest that head-head/head-tail interactions have been conserved, with slight modifications, as a mechanism for regulating myosin II activity from the emergence of the first animals and before. The early origins of these interactions highlight their importance in generating the inhibited (relaxed) state of myosin in muscle and nonmuscle cells.
Keywords: evolution; interacting-heads motif; muscle; myosin II; myosin regulation.
Conflict of interest statement
The authors declare no conflict of interest.
Figures




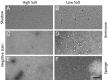
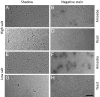

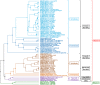
References
-
- Sellers JR. Myosins. 2nd Ed Oxford Univ Press; New York: 1999.
-
- Craig R, Woodhead JL. Structure and function of myosin filaments. Curr Opin Struct Biol. 2006;16:204–212. - PubMed
-
- Huxley HE, Brown W. The low-angle X-ray diagram of vertebrate striated muscle and its behaviour during contraction and rigor. J Mol Biol. 1967;30:383–434. - PubMed
-
- Huxley HE. Structural difference between resting and rigor muscle; evidence from intensity changes in the low-angle equatorial X-ray diagram. J Mol Biol. 1968;37:507–520. - PubMed
-
- Cross RA. What is 10S myosin for? J Muscle Res Cell Motil. 1988;9:108–110. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

