Evaluation of a New Noninvasive Glucose Monitoring Device by Means of Standardized Meal Experiments
- PMID: 29451016
- PMCID: PMC6232728
- DOI: 10.1177/1932296818758769
Evaluation of a New Noninvasive Glucose Monitoring Device by Means of Standardized Meal Experiments
Abstract
Background: Frequent blood glucose readings are the most cumbersome aspect of diabetes treatment for many patients. The noninvasive TensorTip Combo Glucometer (CoG) component employs dedicated mathematical algorithms to analyze the collected signal and to predict tissue glucose at the fingertip. This study presents the performance of the CoG (the invasive and the noninvasive components) during a standardized meal experiment.
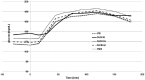
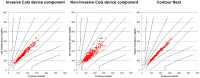
Methods: Each of the 36 participants (18 females and males each, age: 49 ± 18 years, 14 healthy subjects, 6 type 1 and 16 type 2 patients) received a device for conducting calibration at home. Thereafter, they ingested a standardized meal. Blood glucose was assessed from capillary blood samples by means of the (non)invasive device, YSI Stat 2300 plus, Contour Next at time points -30, 0, 15, 30, 45, 60, 75, 90, 120, 150, and 180 minutes. Statistical analysis was performed by consensus error grid (CEG) and calculation of mean absolute relative difference (MARD) in comparison to YSI.
Results: For the noninvasive (NI) CoG technology, 100% of the data pairs were found in CEG zones A (96.6%) and B (3.4%); 100% were seen in zone A for the invasive component and Contour Next. MARD was calculated to be 4.2% for Contour Next, 9.2% for the invasive component, and 14.4% for the NI component.
Conclusions: After appropriate individual calibration of the NI technology, both the NI and the invasive CoG components reliably tracked tissue and blood glucose values, respectively. This may enable patients with diabetes to monitor their glucose levels frequently, reliably, and most of all pain-free.
Keywords: chaos theory; color sensor imaging; fingertip tissue; invasive device component; noninvasive glucose prediction.
Conflict of interest statement
Figures

Similar articles
-
System Accuracy Assessment of a Combined Invasive and Noninvasive Glucometer.J Diabetes Sci Technol. 2020 May;14(3):575-581. doi: 10.1177/1932296819883306. Epub 2019 Oct 22. J Diabetes Sci Technol. 2020. PMID: 31640424 Free PMC article.
-
Device and Method for Noninvasive Glucose Assessment.J Diabetes Sci Technol. 2018 Nov;12(6):1159-1168. doi: 10.1177/1932296818763457. Epub 2018 Mar 24. J Diabetes Sci Technol. 2018. PMID: 29575926 Free PMC article.
-
Noninvasive glucose monitoring: increasing accuracy by combination of multi-technology and multi-sensors.J Diabetes Sci Technol. 2010 May 1;4(3):583-95. doi: 10.1177/193229681000400312. J Diabetes Sci Technol. 2010. PMID: 20513324 Free PMC article. Clinical Trial.
-
A Perspective on the Accuracy of Blood Glucose Meters During Pregnancy.Diabetes Care. 2018 Oct;41(10):2053-2058. doi: 10.2337/dc18-0833. Diabetes Care. 2018. PMID: 30237233 Review.
-
Is it possible to constantly and accurately monitor blood sugar levels, in people with Type 1 diabetes, with a discrete device (non-invasive or invasive)?Diabet Med. 2020 Apr;37(4):532-544. doi: 10.1111/dme.13942. Epub 2019 Mar 13. Diabet Med. 2020. PMID: 30803028 Review.
Cited by
-
Review of Non-invasive Glucose Sensing Techniques: Optical, Electrical and Breath Acetone.Sensors (Basel). 2020 Feb 25;20(5):1251. doi: 10.3390/s20051251. Sensors (Basel). 2020. PMID: 32106464 Free PMC article. Review.
-
Guidelines and Recommendations for Laboratory Analysis in the Diagnosis and Management of Diabetes Mellitus.Diabetes Care. 2023 Oct 1;46(10):e151-e199. doi: 10.2337/dci23-0036. Diabetes Care. 2023. PMID: 37471273 Free PMC article. Review.
-
The Progress of Glucose Monitoring-A Review of Invasive to Minimally and Non-Invasive Techniques, Devices and Sensors.Sensors (Basel). 2019 Feb 15;19(4):800. doi: 10.3390/s19040800. Sensors (Basel). 2019. PMID: 30781431 Free PMC article. Review.
-
System Accuracy Assessment of a Combined Invasive and Noninvasive Glucometer.J Diabetes Sci Technol. 2020 May;14(3):575-581. doi: 10.1177/1932296819883306. Epub 2019 Oct 22. J Diabetes Sci Technol. 2020. PMID: 31640424 Free PMC article.
-
Device and Method for Noninvasive Glucose Assessment.J Diabetes Sci Technol. 2018 Nov;12(6):1159-1168. doi: 10.1177/1932296818763457. Epub 2018 Mar 24. J Diabetes Sci Technol. 2018. PMID: 29575926 Free PMC article.
References
-
- International Diabetes Federation. IDF diabetes atlas— 8th edition. 2017. Available at: www.diabetesatlas.org. - PubMed
-
- UK Prospective Diabetes Study Group. Intensive blood-glucose control with sulphonylureas or insulin compared with conventional treatment and risk of complications in patients with type 2 diabetes (UKPDS 33). Lancet. 1998;352(9131):837-853. - PubMed
-
- Nathan DM, Genuth S, Lachin J, et al. The effect of intensive treatment of diabetes on the development and progression of long-term complications in insulin-dependent diabetes mellitus. N Engl J Med. 1993;329(14):977-986. - PubMed
-
- Karter AJ, Ackerson LM, Darbinian JA, et al. Self-monitoring of blood glucose levels and glycemic control: the Northern California Kaiser Permanente Diabetes registry. Am J Med. 2001;111(1):1-9. - PubMed
-
- Elgart JF, Gonzalez L, Prestes M, Rucci E, Gagliardino JJ. Frequency of self-monitoring blood glucose and attainment of HbA1c target values. Acta Diabetol. 2016;53(1):57-62. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

