Supraspinal modulation of neuronal synchronization by nociceptive stimulation induces an enduring reorganization of dorsal horn neuronal connectivity
- PMID: 29451306
- PMCID: PMC5924834
- DOI: 10.1113/JP275228
Supraspinal modulation of neuronal synchronization by nociceptive stimulation induces an enduring reorganization of dorsal horn neuronal connectivity
Abstract
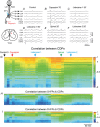
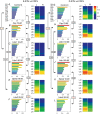
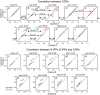
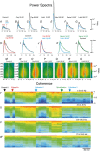
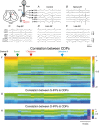
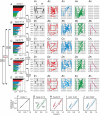
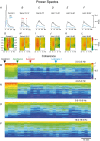
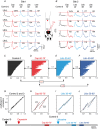
Key points: The state of central sensitization induced by the intradermic injection of capsaicin leads to structured (non-random) changes in functional connectivity between dorsal horn neuronal populations distributed along the spinal lumbar segments in anaesthetized cats. The capsaicin-induced changes in neuronal connectivity and the concurrent increase in secondary hyperalgesia are transiently reversed by the systemic administration of small doses of lidocaine, a clinically effective procedure to treat neuropathic pain. The effects of both capsaicin and lidocaine are greatly attenuated in spinalized preparations, showing that supraspinal influences play a significant role in the shaping of nociceptive-induced changes in dorsal horn functional neuronal connectivity. We conclude that changes in functional connectivity between segmental populations of dorsal horn neurones induced by capsaicin and lidocaine result from a cooperative adaptive interaction between supraspinal and spinal neuronal networks, a process that may have a relevant role in the pathogenesis of chronic pain and analgesia.
Abstract: Despite a profusion of information on the molecular and cellular mechanisms involved in the central sensitization produced by intense nociceptive stimulation, the changes in the patterns of functional connectivity between spinal neurones associated with the development of secondary hyperalgesia and allodynia remain largely unknown. Here we show that the state of central sensitization produced by the intradermal injection of capsaicin is associated with structured transformations in neuronal synchronization that lead to an enduring reorganization of the functional connectivity within a segmentally distributed ensemble of dorsal horn neurones. These changes are transiently reversed by the systemic administration of small doses of lidocaine, a clinically effective procedure to treat neuropathic pain. Lidocaine also reduces the capsaicin-induced facilitation of the spinal responses evoked by weak mechanical stimulation of the skin in the region of secondary but not primary hyperalgesia. The effects of both intradermic capsaicin and systemic lidocaine on the segmental correlation and coherence between ongoing cord dorsum potentials and on the responses evoked by tactile stimulation in the region of secondary hyperalgesia are greatly attenuated in spinalized preparations, showing that supraspinal influences are involved in the reorganization of the nociceptive-induced structured patterns of dorsal horn neuronal connectivity. We conclude that the structured reorganization of the functional connectivity between the dorsal horn neurones induced by capsaicin nociceptive stimulation results from cooperative interactions between supraspinal and spinal networks, a process that may have a relevant role in the shaping of the spinal state in the pathogenesis of chronic pain and analgesia.
Keywords: analgesia; capsaicin; lidocaine; nociception; spinal cord.
© 2018 The Authors. The Journal of Physiology © 2018 The Physiological Society.
Figures




References
-
- Abarbanel HDI, Rabinovich MI, Selverston A, Bazhenov MV, Huerta R, Sushchik MM & Rubchinskii LL (1996). Synchronization in neural networks. Physics‐Uspekhi 39, 337–362.
-
- Averbeck BB & Lee D (2004). Coding and transmission of information by neural ensembles. Trends Neurosci 27, 225–230. - PubMed
-
- Bak P (1997). How Nature Works: The Science of Self‐organized Criticality. Oxford University Press, Oxford, UK.
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

