Increase of 10% in the Rate of Adverse Drug Reactions for Each Drug Administered in Hospitalized Patients
- PMID: 29451619
- PMCID: PMC5784181
- DOI: 10.6061/clinics/2018/e185
Increase of 10% in the Rate of Adverse Drug Reactions for Each Drug Administered in Hospitalized Patients
Abstract
Objective: To assess the risk factors, incidence and severity of adverse drug reactions in in-patients.
Methods: This prospective study evaluated 472 patients treated at a teaching hospital in Brazil between 2010 and 2013 by five medical specialties: Internal Medicine, General Surgery, Geriatrics, Neurology, and Clinical Immunology and Allergy. The following variables were assessed: patient age, gender, comorbidities, family history of hypersensitivity, personal and family history of atopy, number of prescribed drugs before and during hospitalization, hospital diagnoses, days of hospitalization. The patients were visited every other day, and medical records were reviewed by the investigators to detect adverse drug reactions.
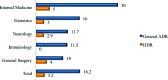
Results: There were a total of 94 adverse drug reactions in 75 patients. Most reactions were predictable and of moderate severity. The incidence of adverse drug reactions was 16.2%, and the incidence varied, according to the medical specialty; it was higher in Internal Medicine (30%). Antibiotics were the most commonly involved medication. Chronic renal failure, longer hospital stay, greater number of diagnoses and greater number of medications upon admission were risk factors. For each medication introduced during hospitalization, there was a 10% increase in the rate of adverse drug reaction. In the present study, the probability of observing an adverse drug reaction was 1 in 104 patients per day.
Conclusions: Adverse drug reactions are frequent and potentially serious and should be better monitored in patients with chronic renal failure or prolonged hospitalization and especially in those on 'polypharmacy' regimens. The rational use of medications plays an important role in preventing adverse drug reactions.
Conflict of interest statement
No potential conflict of interest was reported.
Figures
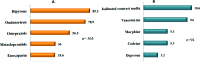
Similar articles
-
Impact of pharmacovigilance on adverse drug reactions reporting in hospitalized internal medicine patients at Saudi Arabian teaching hospital.Saudi Med J. 2012 Aug;33(8):863-8. Saudi Med J. 2012. PMID: 22886119
-
Prevalence, risk factors and main features of adverse drug reactions leading to hospital admission.Eur J Clin Pharmacol. 2014 Mar;70(3):361-7. doi: 10.1007/s00228-013-1630-5. Epub 2013 Dec 21. Eur J Clin Pharmacol. 2014. PMID: 24362489
-
Adverse Drug Reactions in an Oncological Population: Prevalence, Predictability, and Preventability.Oncologist. 2019 Sep;24(9):e968-e977. doi: 10.1634/theoncologist.2018-0476. Epub 2019 Mar 4. Oncologist. 2019. PMID: 30833488 Free PMC article.
-
Epidemiology of drug exposure and adverse drug reactions in two swiss departments of internal medicine.Br J Clin Pharmacol. 2000 Feb;49(2):158-67. doi: 10.1046/j.1365-2125.2000.00132.x. Br J Clin Pharmacol. 2000. PMID: 10671911 Free PMC article.
-
Risk Factors Associated With Drug-Related Side Effects in Children: A Scoping Review.Glob Pediatr Health. 2024 Aug 27;11:2333794X241273171. doi: 10.1177/2333794X241273171. eCollection 2024. Glob Pediatr Health. 2024. PMID: 39205860 Free PMC article.
Cited by
-
The Psychological Relieving Effect of Drug Provocation Test in Drug Hypersensitivity.J Asthma Allergy. 2022 Sep 24;15:1337-1345. doi: 10.2147/JAA.S380516. eCollection 2022. J Asthma Allergy. 2022. PMID: 36189190 Free PMC article.
-
Astragalus polysaccharide (PG2) Ameliorates Cancer Symptom Clusters, as well as Improves Quality of Life in Patients with Metastatic Disease, through Modulation of the Inflammatory Cascade.Cancers (Basel). 2019 Jul 25;11(8):1054. doi: 10.3390/cancers11081054. Cancers (Basel). 2019. PMID: 31349728 Free PMC article.
-
Identification of potentially causative drugs associated with hypotension: A scoping review.Arch Pharm (Weinheim). 2025 Jan;358(1):e2400564. doi: 10.1002/ardp.202400564. Epub 2024 Nov 28. Arch Pharm (Weinheim). 2025. PMID: 39607387 Free PMC article.
-
Real-World Complexity of Prescribing Cascades.Basic Clin Pharmacol Toxicol. 2025 Jul;137(1):e70063. doi: 10.1111/bcpt.70063. Basic Clin Pharmacol Toxicol. 2025. PMID: 40457590 Free PMC article. Review.
-
A review on adverse drug reaction related to medication in health sector: an account of what we have discovered and implemented-pharmacovigilance.Naunyn Schmiedebergs Arch Pharmacol. 2025 May 19. doi: 10.1007/s00210-025-04262-0. Online ahead of print. Naunyn Schmiedebergs Arch Pharmacol. 2025. PMID: 40387927 Review.
References
-
- Johansson SG, Bieber T, Dahl R, Friedmann PS, Lanier BQ, Lockey RF, et al. Revised nomenclature for allergy for global use: Report of the Nomenclature Review Committee of the World Allergy Organization, October 2003. J Allergy Clin Immunol. 2004;113((5)):832–6. doi: 10.1016/j.jaci.2003.12.591. - DOI - PubMed
-
- Kvasz M, Allen IE, Gordon MJ, Ro EY, Estok R, Olkin I, et al. Adverse drug reactions in hospitalized patients: A critique of a meta-analysis. MedGenMed. 2000;2((2)):E3. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

