Acute Pressor Response to Psychosocial Stress Is Dependent on Endothelium-Derived Endothelin-1
- PMID: 29453306
- PMCID: PMC5850198
- DOI: 10.1161/JAHA.117.007863
Acute Pressor Response to Psychosocial Stress Is Dependent on Endothelium-Derived Endothelin-1
Abstract
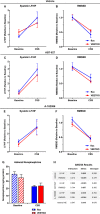
Background: Acute psychosocial stress provokes increases in circulating endothelin-1 (ET-1) levels in humans and animal models. However, key questions about the physiological function and cellular source of stress-induced ET-1 remain unanswered. We hypothesized that endothelium-derived ET-1 contributes to the acute pressor response to stress via activation of the endothelin A receptor.
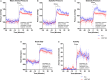
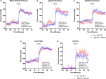
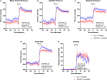
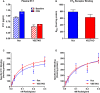
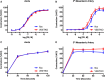
Methods and results: Adult male vascular endothelium-specific ET-1 knockout mice and control mice that were homozygous for the floxed allele were exposed to acute psychosocial stress in the form of cage switch stress (CSS), with blood pressure measured by telemetry. An acute pressor response was elicited by CSS in both genotypes; however, this response was significantly blunted in vascular endothelium-specific ET-1 knockout mice compared with control mice that were homozygous for the floxed allele. In mice pretreated for 3 days with the endothelin A antagonist, ABT-627, or the dual endothelin A/B receptor antagonist, A-182086, the pressor response to CSS was similar between genotypes. CSS significantly increased plasma ET-1 levels in control mice that were homozygous for the floxed allele. CSS failed to elicit an increase in plasma ET-1 in vascular endothelium-specific ET-1 knockout mice. Telemetry frequency domain analyses suggested similar autonomic responses to stress between genotypes, and isolated resistance arteries demonstrated similar sensitivity to α1-adrenergic receptor-mediated vasoconstriction.
Conclusions: These findings specify that acute stress-induced activation of endothelium-derived ET-1 and subsequent endothelin A receptor activation is a novel mediator of the blood pressure response to acute psychosocial stress.
Keywords: cage switch stress; endothelin‐1; endothelium‐derived factors; psychosocial stress; stress; vascular endothelium‐specific ET‐1 knockout mice.
© 2018 The Authors. Published on behalf of the American Heart Association, Inc., by Wiley.
Figures
References
-
- McEwen BS. Physiology and neurobiology of stress and adaptation: central role of the brain. Physiol Rev. 2007;87:873–904. - PubMed
-
- Treiber FA, Kapuku GK, Davis H, Pollock JS, Pollock DM. Plasma endothelin‐1 release during acute stress: role of ethnicity and sex. Psychosom Med. 2002;64:707–713. - PubMed
-
- Lee DL, Webb RC, Brands MW. Sympathetic and angiotensin‐dependent hypertension during cage‐switch stress in mice. Am J Physiol Regul Integr Comp Physiol. 2004;287:R1394–R1398. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

