Distinct CD40L receptors mediate inflammasome activation and secretion of IL-1β and MCP-1 in cultured human retinal pigment epithelial cells
- PMID: 29454857
- PMCID: PMC5924621
- DOI: 10.1016/j.exer.2018.02.014
Distinct CD40L receptors mediate inflammasome activation and secretion of IL-1β and MCP-1 in cultured human retinal pigment epithelial cells
Abstract
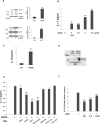
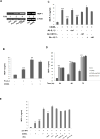
CD40L signaling occurs in several diseases with inflammatory components, including ocular and retinal diseases. However, it has never been evaluated as a pathogenic mechanism in age-related macular degeneration (AMD) or as an inducer of inflammasome formation in any cell type. mRNA and protein levels of CD40, IL-1β, NALP1, NALP3, caspase-1, and caspase-5 were determined by RT-PCR, qPCR, and Western blot. CD40L receptor (CD40, α5β1, and CD11b) expression was determined by Western and immunofluorescent staining. IL-1β, IL-18, and MCP-1 secretions were determined by ELISA. NALP1 and NALP3 inflammasome formation were determined by Co-IP. Experiments were conducted on primary human retinal pigment epithelial (hRPE) cells from four different donors. Human umbilical vein endothelial (HUVEC) and monocytic leukemia (THP-1) cells demonstrated the general applicability of our findings. In hRPE cells, CD40L-induced NALP1 and NALP3 inflammasome activation, cleavage of caspase-1 and caspase-5, and IL-1β and IL-18 secretion. Interestingly, neutralizing CD11b and α5β1 antibodies, but not CD40, reduced CD40L-induced IL-1β secretion in hRPE cells. Similarly, CD40L treatment also induced HUVEC and THP-1 cells to secret IL-1β through CD11b and α5β1. Additionally, the CD40L-induced IL-1β secretion acted in an autocrine/paracrine manner to feed back and induce hRPE cells to secrete MCP-1. This study is the first to show that CD40L induces inflammasome activation in any cell type, including hRPE cells, and that this induction is through CD11b and α5β1 cell-surface receptors. These mechanisms likely play an important role in many retinal and non-retinal diseases and provide compelling drug targets that may help reduce pro-inflammatory processes.
Keywords: AMD; Age-related macular degeneration; CD40L; IL-1β; Inflammasome; MCP-1; RPE; Retinal pigment epithelium.
Copyright © 2018 Elsevier Ltd. All rights reserved.
Figures




Similar articles
-
Interleukin-17A Induces IL-1β Secretion From RPE Cells Via the NLRP3 Inflammasome.Invest Ophthalmol Vis Sci. 2016 Feb;57(2):312-9. doi: 10.1167/iovs.15-17578. Invest Ophthalmol Vis Sci. 2016. PMID: 26830368
-
Expression and regulation of alarmin cytokine IL-1α in human retinal pigment epithelial cells.Exp Eye Res. 2018 Jul;172:10-20. doi: 10.1016/j.exer.2018.03.015. Epub 2018 Mar 15. Exp Eye Res. 2018. PMID: 29551335 Free PMC article.
-
Fluorofenidone attenuates pulmonary inflammation and fibrosis via inhibiting the activation of NALP3 inflammasome and IL-1β/IL-1R1/MyD88/NF-κB pathway.J Cell Mol Med. 2016 Nov;20(11):2064-2077. doi: 10.1111/jcmm.12898. Epub 2016 Jun 16. J Cell Mol Med. 2016. PMID: 27306439 Free PMC article.
-
Contribution of inflammasome complex in inflammatory-related eye disorders and its implications for anti-inflammasome therapy.Immunopharmacol Immunotoxicol. 2020 Oct;42(5):400-407. doi: 10.1080/08923973.2020.1808986. Epub 2020 Aug 29. Immunopharmacol Immunotoxicol. 2020. PMID: 32791926 Review.
-
IL-1 Family Members Mediate Cell Death, Inflammation and Angiogenesis in Retinal Degenerative Diseases.Front Immunol. 2019 Jul 16;10:1618. doi: 10.3389/fimmu.2019.01618. eCollection 2019. Front Immunol. 2019. PMID: 31379825 Free PMC article. Review.
Cited by
-
Differential induction of donor-reactive Foxp3+ regulatory T cell via blockade of CD154 vs CD40.Am J Transplant. 2024 Aug;24(8):1369-1381. doi: 10.1016/j.ajt.2024.03.033. Epub 2024 Mar 27. Am J Transplant. 2024. PMID: 38552961 Free PMC article.
-
Methyltransferase-like (METTL)14-mediated N6-methyladenosine modification modulates retinal pigment epithelial (RPE) activity by regulating the methylation of microtubule-associated protein (MAP)2.Bioengineered. 2022 Mar;13(3):4773-4785. doi: 10.1080/21655979.2022.2032968. Bioengineered. 2022. PMID: 35139773 Free PMC article.
-
Ainsliadimer C, a disesquiterpenoid isolated from Ainsliaea macrocephala, ameliorates inflammatory responses in adipose tissue via Sirtuin 1-NLRP3 inflammasome axis.Acta Pharmacol Sin. 2022 Jul;43(7):1780-1792. doi: 10.1038/s41401-021-00797-z. Epub 2021 Nov 17. Acta Pharmacol Sin. 2022. PMID: 34789920 Free PMC article.
-
The CD40-CD40L Dyad as Immunotherapeutic Target in Cardiovascular Disease.J Cardiovasc Transl Res. 2021 Feb;14(1):13-22. doi: 10.1007/s12265-020-09994-3. Epub 2020 Mar 28. J Cardiovasc Transl Res. 2021. PMID: 32222950 Free PMC article. Review.
-
PGC-1α Protects RPE Cells of the Aging Retina against Oxidative Stress-Induced Degeneration through the Regulation of Senescence and Mitochondrial Quality Control. The Significance for AMD Pathogenesis.Int J Mol Sci. 2018 Aug 7;19(8):2317. doi: 10.3390/ijms19082317. Int J Mol Sci. 2018. PMID: 30087287 Free PMC article. Review.
References
-
- Andre P, Nannizzi-Alaimo L, Prasad SK, Phillips DR. Platelet-derived CD40L: the switch-hitting player of cardiovascular disease. Circulation. 2002;106:896–899. - PubMed
-
- Bagenstose LM, Agarwal RK, Silver PB, Harlan DM, Hoffmann SC, Kampen RL, Chan CC, Caspi RR. Disruption of CD40/CD40-ligand interactions in a retinal autoimmunity model results in protection without tolerance. J Immunol. 2005;175:124–130. - PubMed
-
- Bian ZM, Elner SG, Yoshida A, Elner VM. Human RPE-monocyte co-culture induces chemokine gene expression through activation of MAPK and NIK cascade. Experimental eye research. 2003;76:573–583. - PubMed
-
- Bian ZM, Elner SG, Yoshida A, Elner VM. Differential involvement of phosphoinositide 3-kinase/Akt in human RPE MCP-1 and IL-8 expression. Investigative ophthalmology & visual science. 2004;45:1887–1896. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

