Extracellular ATP is Differentially Metabolized on Papillary Thyroid Carcinoma Cells Surface in Comparison to Normal Cells
- PMID: 29455338
- PMCID: PMC6008267
- DOI: 10.1007/s12307-018-0206-4
Extracellular ATP is Differentially Metabolized on Papillary Thyroid Carcinoma Cells Surface in Comparison to Normal Cells
Abstract
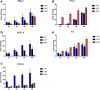
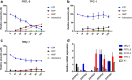
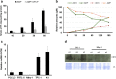
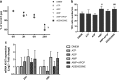
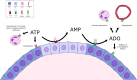
The incidence of differentiated thyroid cancer has been increasing. Nevertheless, its molecular mechanisms are not well understood. In recent years, extracellular nucleotides and nucleosides have emerged as important modulators of tumor microenvironment. Extracellular ATP is mainly hydrolyzed by NTPDase1/CD39 and NTPDase2/CD39L1, generating AMP, which is hydrolyzed by ecto-5'-nucleotidase (CD73) to adenosine, a possible promoter of tumor growth and metastasis. There are no studies evaluating the expression and functionality of these ectonucleotidases on normal or tumor-derived thyroid cells. Thus, we investigated the ability of thyroid cancer cells to hydrolyze extracellular ATP generating adenosine, and the expression of ecto-enzymes, as compared to normal cells. We found that normal thyroid derived cells presented a higher ability to hydrolyze ATP and higher mRNA levels for ENTDP1-2, when compared to papillary thyroid carcinoma (PTC) derived cells, which had a higher ability to hydrolyze AMP and expressed CD73 mRNA and protein at higher levels. In addition, adenosine induced an increase in proliferation and migration in PTC derived cells, whose effect was blocked by APCP, a non-hydrolysable ADP analogue, which is an inhibitor of CD73. Taken together, these results showed that thyroid follicular cells have a functional purinergic signaling. The higher expression of CD73 in PTC derived cells might favor the accumulation of extracellular adenosine in the tumor microenvironment, which could promote tumor progression. Therefore, as already shown for other tumors, the purinergic signaling should be considered a potential target for thyroid cancer management and treatment.
Keywords: Adenosine; CD73; Extracellular ATP; NTPDases; Purinergic signaling; Thyroid papillary carcinoma.
Conflict of interest statement
No conflicts of interest, financial or otherwise, are declared by the authors.
Figures
Similar articles
-
Alterations in the extracellular catabolism of nucleotides are involved in the antiproliferative effect of quercetin in human bladder cancer T24 cells.Urol Oncol. 2013 Oct;31(7):1204-11. doi: 10.1016/j.urolonc.2011.10.009. Epub 2011 Dec 3. Urol Oncol. 2013. PMID: 22137869
-
Activity of ecto-5'-nucleotidase (NT5E/CD73) is increased in papillary thyroid carcinoma and its expression is associated with metastatic lymph nodes.Mol Cell Endocrinol. 2019 Jan 5;479:54-60. doi: 10.1016/j.mce.2018.08.013. Epub 2018 Sep 2. Mol Cell Endocrinol. 2019. PMID: 30184475
-
Epidermal melanocytes metabolize extracellular nucleotides by purinergic enzymes.Biochem Cell Biol. 2023 Jun 1;101(3):259-266. doi: 10.1139/bcb-2022-0058. Epub 2023 Jan 19. Biochem Cell Biol. 2023. PMID: 36657128
-
Purinergic Signaling in Glioma Progression.Adv Exp Med Biol. 2020;1202:87-108. doi: 10.1007/978-3-030-30651-9_5. Adv Exp Med Biol. 2020. PMID: 32034710 Review.
-
Multifaceted Effects of Extracellular Adenosine Triphosphate and Adenosine in the Tumor-Host Interaction and Therapeutic Perspectives.Front Immunol. 2017 Nov 14;8:1526. doi: 10.3389/fimmu.2017.01526. eCollection 2017. Front Immunol. 2017. PMID: 29184552 Free PMC article. Review.
Cited by
-
Review immune response of targeting CD39 in cancer.Biomark Res. 2023 Jun 7;11(1):63. doi: 10.1186/s40364-023-00500-w. Biomark Res. 2023. PMID: 37287049 Free PMC article. Review.
-
CD73 expression in normal, hyperplastic, and neoplastic thyroid: a systematic evaluation revealing CD73 overexpression as a feature of papillary carcinomas.Virchows Arch. 2021 Jul;479(1):209-214. doi: 10.1007/s00428-021-03100-x. Epub 2021 May 21. Virchows Arch. 2021. PMID: 34019179 Free PMC article.
-
ATPergic signaling disruption in human sepsis as a potential source of biomarkers for clinical use.Clin Exp Med. 2023 Nov;23(7):3651-3662. doi: 10.1007/s10238-023-01045-w. Epub 2023 Mar 21. Clin Exp Med. 2023. PMID: 36943594 Free PMC article.
-
Imatinib mesylate affects extracellular ATP catabolism and expression of NTPDases in a chronic myeloid leukemia cell line.Purinergic Signal. 2020 Mar;16(1):29-40. doi: 10.1007/s11302-019-09686-x. Epub 2020 Jan 18. Purinergic Signal. 2020. PMID: 31955347 Free PMC article.
-
CD73 mitigates ZEB1 expression in papillary thyroid carcinoma.Cell Commun Signal. 2024 Feb 22;22(1):145. doi: 10.1186/s12964-024-01522-z. Cell Commun Signal. 2024. PMID: 38388432 Free PMC article.
References
Grants and funding
- Programa Nacional de Pós-Doutorado (PNPD)/Coordenação de Aperfeiçoamento de Pessoal de Nível Superior
- PROBITEC (004/2012)/Coordenação de Aperfeiçoamento de Pessoal de Nível Superior
- PROCAD (158819)/Coordenação de Aperfeiçoamento de Pessoal de Nível Superior
- Research fellowship/Conselho Nacional de Desenvolvimento Científico e Tecnológico
- Research fellowship/Conselho Nacional de Desenvolvimento Científico e Tecnológico
- Research fellowship/Conselho Nacional de Desenvolvimento Científico e Tecnológico
- Novas Terapias Portadoras de Futuro (457394/2013-7)/Conselho Nacional de Desenvolvimento Científico e Tecnológico
- ICGEB (405231/2015-6 MCTI/CNPq-ICGEB)/Conselho Nacional de Desenvolvimento Científico e Tecnológico
- Pronex FAPERGS/CNPq 16/2551-0000473-0)/Fundação de Amparo à Pesquisa do Estado do Rio Grande do Sul
- FIPE/HCPA (Nº 15-0590)/Hospital de Clínicas de Porto Alegre
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

