Mechanical Division of Cell-Sized Liposomes
- PMID: 29455527
- PMCID: PMC5876618
- DOI: 10.1021/acsnano.7b08411
Mechanical Division of Cell-Sized Liposomes
Abstract
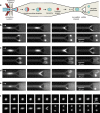
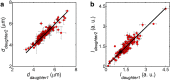
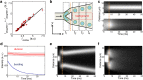
Liposomes, self-assembled vesicles with a lipid-bilayer boundary similar to cell membranes, are extensively used in both fundamental and applied sciences. Manipulation of their physical properties, such as growth and division, may significantly expand their use as model systems in cellular and synthetic biology. Several approaches have been explored to controllably divide liposomes, such as shape transformation through temperature cycling, incorporation of additional lipids, and the encapsulation of protein division machinery. However, so far, these methods lacked control, exhibited low efficiency, and yielded asymmetric division in terms of volume or lipid composition. Here, we present a microfluidics-based strategy to realize mechanical division of cell-sized (∼6 μm) liposomes. We use octanol-assisted liposome assembly (OLA) to produce liposomes on chip, which are subsequently flowed against the sharp edge of a wedge-shaped splitter. Upon encountering such a Y-shaped bifurcation, the liposomes are deformed and, remarkably, are able to divide into two stable daughter liposomes in just a few milliseconds. The probability of successful division is found to critically depend on the surface area-to-volume ratio of the mother liposome, which can be tuned through osmotic pressure, and to strongly correlate to the mother liposome size for given microchannel dimensions. The division process is highly symmetric (∼3% size variation between the daughter liposomes) and is accompanied by a low leakage. This mechanical division of liposomes may constitute a valuable step to establish a growth-division cycle of synthetic cells.
Keywords: liposomes; membrane biophysics; microfluidics; octanol-assisted liposome assembly; synthetic biology; synthetic cell division.
Conflict of interest statement
The authors declare no competing financial interest.
Figures

References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

