Staphylococcal Protein A Is a Key Factor in Neutrophil Extracellular Traps Formation
- PMID: 29459871
- PMCID: PMC5807341
- DOI: 10.3389/fimmu.2018.00165
Staphylococcal Protein A Is a Key Factor in Neutrophil Extracellular Traps Formation
Abstract
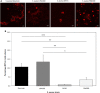
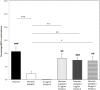
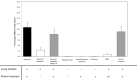
Staphylococcus aureus are strong inducers of neutrophil extracellular traps (NETs), a defense mechanism of neutrophils against pathogens. Our aim was to explore the role of Protein A in S. aureus-induced NETosis. We determined the Protein A production of four different S. aureus strains and found a direct relationship between the degree of NETosis induction and Protein A production: strains producing higher concentrations of Protein A evoke significantly more NETs. A S. aureus strain in which Protein A as well as a second binding protein for immunoglobulins (Sbi) have been knocked-out (ΔSpA ΔSbi) induced significantly less NETosis than the wild-type strain. NETosis induction by this knockout strain can be rescued by the addition of purified Protein A. Dead S. aureus did not induce NETosis. In conclusion, Protein A is a determinant for NETosis induction by S. aureus.
Keywords: NETs; Protein A; S. aureus; SpA; Staphylococcus aureus; neutrophil extracellular traps.
Figures


Similar articles
-
Staphylococcus aureus and Neutrophil Extracellular Traps: The Master Manipulator Meets Its Match in Immunothrombosis.Arterioscler Thromb Vasc Biol. 2022 Mar;42(3):261-276. doi: 10.1161/ATVBAHA.121.316930. Epub 2022 Feb 3. Arterioscler Thromb Vasc Biol. 2022. PMID: 35109674 Free PMC article. Review.
-
Calcium depletion at high glucose concentration promotes vesicle-mediated NET release in response to Staphylococcus aureus.Mol Immunol. 2020 Aug;124:211-217. doi: 10.1016/j.molimm.2020.06.015. Epub 2020 Jun 27. Mol Immunol. 2020. PMID: 32603960
-
Triggering NETosis via protease-activated receptor (PAR)-2 signaling as a mechanism of hijacking neutrophils function for pathogen benefits.PLoS Pathog. 2019 May 20;15(5):e1007773. doi: 10.1371/journal.ppat.1007773. eCollection 2019 May. PLoS Pathog. 2019. PMID: 31107907 Free PMC article.
-
During the Early Stages of Staphylococcus aureus Biofilm Formation, Induced Neutrophil Extracellular Traps Are Degraded by Autologous Thermonuclease.Infect Immun. 2019 Nov 18;87(12):e00605-19. doi: 10.1128/IAI.00605-19. Print 2019 Dec. Infect Immun. 2019. PMID: 31527127 Free PMC article.
-
Molecular Prerequisites for Neutrophil Extracellular Trap Formation and Evasion Mechanisms of Staphylococcus aureus.Front Immunol. 2022 Feb 14;13:836278. doi: 10.3389/fimmu.2022.836278. eCollection 2022. Front Immunol. 2022. PMID: 35237275 Free PMC article. Review.
Cited by
-
An In Vitro Study of the Effect of Viburnum opulus Extracts on Key Processes in the Development of Staphylococcal Infections.Molecules. 2021 Mar 21;26(6):1758. doi: 10.3390/molecules26061758. Molecules. 2021. PMID: 33801012 Free PMC article.
-
Staphylococcus aureus and Neutrophil Extracellular Traps: The Master Manipulator Meets Its Match in Immunothrombosis.Arterioscler Thromb Vasc Biol. 2022 Mar;42(3):261-276. doi: 10.1161/ATVBAHA.121.316930. Epub 2022 Feb 3. Arterioscler Thromb Vasc Biol. 2022. PMID: 35109674 Free PMC article. Review.
-
Binding of Staphylococcus aureus Protein A to von Willebrand Factor Is Regulated by Mechanical Force.mBio. 2019 Apr 30;10(2):e00555-19. doi: 10.1128/mBio.00555-19. mBio. 2019. PMID: 31040240 Free PMC article.
-
Case report: In vivo detection of neutrophil extracellular traps in a dog with thrombosis induced by bacterial vasculitis.Front Vet Sci. 2025 Feb 12;12:1470605. doi: 10.3389/fvets.2025.1470605. eCollection 2025. Front Vet Sci. 2025. PMID: 40012748 Free PMC article.
-
Paracoccidioides brasiliensis Releases a DNase-Like Protein That Degrades NETs and Allows for Fungal Escape.Front Cell Infect Microbiol. 2021 Feb 10;10:592022. doi: 10.3389/fcimb.2020.592022. eCollection 2020. Front Cell Infect Microbiol. 2021. PMID: 33643928 Free PMC article.
References
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

