RNF8 and SCML2 cooperate to regulate ubiquitination and H3K27 acetylation for escape gene activation on the sex chromosomes
- PMID: 29462142
- PMCID: PMC5834201
- DOI: 10.1371/journal.pgen.1007233
RNF8 and SCML2 cooperate to regulate ubiquitination and H3K27 acetylation for escape gene activation on the sex chromosomes
Abstract
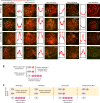
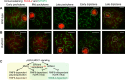
The sex chromosomes are enriched with germline genes that are activated during the late stages of spermatogenesis. Due to meiotic sex chromosome inactivation (MSCI), these sex chromosome-linked genes must escape silencing for activation in spermatids, thereby ensuring their functions for male reproduction. RNF8, a DNA damage response protein, and SCML2, a germline-specific Polycomb protein, are two major, known regulators of this process. Here, we show that RNF8 and SCML2 cooperate to regulate ubiquitination during meiosis, an early step to establish active histone modifications for subsequent gene activation. Double mutants of Rnf8 and Scml2 revealed that RNF8-dependent monoubiquitination of histone H2A at Lysine 119 (H2AK119ub) is deubiquitinated by SCML2, demonstrating interplay between RNF8 and SCML2 in ubiquitin regulation. Additionally, we identify distinct functions of RNF8 and SCML2 in the regulation of ubiquitination: SCML2 deubiquitinates RNF8-independent H2AK119ub but does not deubiquitinate RNF8-dependent polyubiquitination. RNF8-dependent polyubiquitination is required for the establishment of H3K27 acetylation, a marker of active enhancers, while persistent H2AK119ub inhibits establishment of H3K27 acetylation. Following the deposition of H3K27 acetylation, H3K4 dimethylation is established as an active mark on poised promoters. Together, we propose a model whereby regulation of ubiquitin leads to the organization of poised enhancers and promoters during meiosis, which induce subsequent gene activation from the otherwise silent sex chromosomes in postmeiotic spermatids.
Conflict of interest statement
AB is a co-founder of Datirium, LLC.
Figures






References
-
- Silber SJ. A modern view of male infertility. Reprod Fertil Dev. 1994;6(1):93–103; discussion -4. Epub 1994/01/01. . - PubMed
-
- Krausz C, Escamilla AR, Chianese C. Genetics of male infertility: from research to clinic. Reproduction (Cambridge, England). 2015;150(5):R159–74. Epub 2015/10/09. doi: 10.1530/rep-15-0261 . - DOI - PubMed
-
- Hu YC, Namekawa SH. Functional significance of the sex chromosomes during spermatogenesis. Reproduction. 2015;149(6):R265–77. Epub 2015/05/08. doi: 10.1530/REP-14-0613 ; PubMed Central PMCID: PMCPMC4510947. - DOI - PMC - PubMed
-
- Luconi M, Cantini G, Baldi E, Forti G. Role of a-kinase anchoring proteins (AKAPs) in reproduction. Front Biosci. 2011;16:1315–30. Epub 2011/01/05. doi: 3791 [pii]. . - PubMed
-
- Lee S, Lee SH, Chung TG, Kim HJ, Yoon TK, Kwak IP, et al. Molecular and cytogenetic characterization of two azoospermic patients with X-autosome translocation. J Assist Reprod Genet. 2003;20(9):385–9. Epub 2003/10/09. doi: 10.1023/A:1025437329427 . - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

