Mechanotransduction in tumor progression: The dark side of the force
- PMID: 29467174
- PMCID: PMC5940296
- DOI: 10.1083/jcb.201701039
Mechanotransduction in tumor progression: The dark side of the force
Abstract
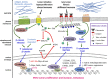
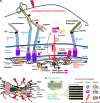
Cancer has been characterized as a genetic disease, associated with mutations that cause pathological alterations of the cell cycle, adhesion, or invasive motility. Recently, the importance of the anomalous mechanical properties of tumor tissues, which activate tumorigenic biochemical pathways, has become apparent. This mechanical induction in tumors appears to consist of the destabilization of adult tissue homeostasis as a result of the reactivation of embryonic developmental mechanosensitive pathways in response to pathological mechanical strains. These strains occur in many forms, for example, hypervascularization in late tumors leads to high static hydrodynamic pressure that can promote malignant progression through hypoxia or anomalous interstitial liquid and blood flow. The high stiffness of tumors directly induces the mechanical activation of biochemical pathways enhancing the cell cycle, epithelial-mesenchymal transition, and cell motility. Furthermore, increases in solid-stress pressure associated with cell hyperproliferation activate tumorigenic pathways in the healthy epithelial cells compressed by the neighboring tumor. The underlying molecular mechanisms of the translation of a mechanical signal into a tumor inducing biochemical signal are based on mechanically induced protein conformational changes that activate classical tumorigenic signaling pathways. Understanding these mechanisms will be important for the development of innovative treatments to target such mechanical anomalies in cancer.
© 2018 Broders-Bondon et al.
Figures


References
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

