Identification of Intestinal UDP-Glucuronosyltransferase Inhibitors in Green Tea (Camellia sinensis) Using a Biochemometric Approach: Application to Raloxifene as a Test Drug via In Vitro to In Vivo Extrapolation
- PMID: 29467215
- PMCID: PMC5890833
- DOI: 10.1124/dmd.117.079491
Identification of Intestinal UDP-Glucuronosyltransferase Inhibitors in Green Tea (Camellia sinensis) Using a Biochemometric Approach: Application to Raloxifene as a Test Drug via In Vitro to In Vivo Extrapolation
Abstract
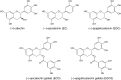
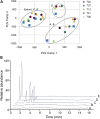
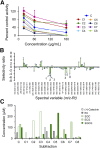
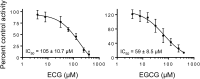
Green tea (Camellia sinensis) is a popular beverage worldwide, raising concern for adverse interactions when co-consumed with conventional drugs. Like many botanical natural products, green tea contains numerous polyphenolic constituents that undergo extensive glucuronidation. As such, the UDP-glucuronosyltransferases (UGTs), particularly intestinal UGTs, represent potential first-pass targets for green tea-drug interactions. Candidate intestinal UGT inhibitors were identified using a biochemometrics approach, which combines bioassay and chemometric data. Extracts and fractions prepared from four widely consumed teas were screened (20-180 μg/ml) as inhibitors of UGT activity (4-methylumbelliferone glucuronidation) in human intestinal microsomes; all demonstrated concentration-dependent inhibition. A biochemometrics-identified fraction rich in UGT inhibitors from a representative tea was purified further and subjected to second-stage biochemometric analysis. Five catechins were identified as major constituents in the bioactive subfractions and prioritized for further evaluation. Of these catechins, (-)-epicatechin gallate and (-)-epigallocatechin gallate showed concentration-dependent inhibition, with IC50 values (105 and 59 μM, respectively) near or below concentrations measured in a cup (240 ml) of tea (66 and 240 μM, respectively). Using the clinical intestinal UGT substrate raloxifene, the Ki values were ∼1.0 and 2.0 μM, respectively. Using estimated intestinal lumen and enterocyte inhibitor concentrations, a mechanistic static model predicted green tea to increase the raloxifene plasma area under the curve up to 6.1- and 1.3-fold, respectively. Application of this novel approach, which combines biochemometrics with in vitro-in vivo extrapolation, to other natural product-drug combinations will refine these procedures, informing the need for further evaluation via dynamic modeling and clinical testing.
Copyright © 2018 by The American Society for Pharmacology and Experimental Therapeutics.
Figures



Similar articles
-
Co-consuming green tea with raloxifene decreases raloxifene systemic exposure in healthy adult participants.Clin Transl Sci. 2023 Oct;16(10):1779-1790. doi: 10.1111/cts.13578. Epub 2023 Aug 28. Clin Transl Sci. 2023. PMID: 37639334 Free PMC article.
-
Identification of diet-derived constituents as potent inhibitors of intestinal glucuronidation.Drug Metab Dispos. 2014 Oct;42(10):1675-83. doi: 10.1124/dmd.114.059451. Epub 2014 Jul 9. Drug Metab Dispos. 2014. PMID: 25008344 Free PMC article.
-
Targeted screen for human UDP-glucuronosyltransferases inhibitors and the evaluation of potential drug-drug interactions with zafirlukast.Drug Metab Dispos. 2015 Jun;43(6):812-8. doi: 10.1124/dmd.114.062141. Epub 2015 Apr 1. Drug Metab Dispos. 2015. PMID: 25834030
-
An Appraisal of Drug-Drug Interactions with Green Tea (Camellia sinensis).Planta Med. 2017 Apr;83(6):496-508. doi: 10.1055/s-0043-100934. Epub 2017 Jan 24. Planta Med. 2017. PMID: 28118673 Review.
-
Green Tea Catechins as Perpetrators of Drug Pharmacokinetic Interactions.Clin Pharmacol Ther. 2025 Jul;118(1):45-61. doi: 10.1002/cpt.3547. Epub 2025 Jan 2. Clin Pharmacol Ther. 2025. PMID: 39748104 Review.
Cited by
-
Developing a Knowledge Graph for Pharmacokinetic Natural Product-Drug Interactions.J Biomed Inform. 2023 Apr;140:104341. doi: 10.1016/j.jbi.2023.104341. Epub 2023 Mar 17. J Biomed Inform. 2023. PMID: 36933632 Free PMC article.
-
Co-consuming green tea with raloxifene decreases raloxifene systemic exposure in healthy adult participants.Clin Transl Sci. 2023 Oct;16(10):1779-1790. doi: 10.1111/cts.13578. Epub 2023 Aug 28. Clin Transl Sci. 2023. PMID: 37639334 Free PMC article.
-
The Importance of Reference Materials and Method Validation for Advancing Research on the Health Effects of Dietary Supplements and Other Natural Products.Front Nutr. 2021 Dec 14;8:786261. doi: 10.3389/fnut.2021.786261. eCollection 2021. Front Nutr. 2021. PMID: 34970578 Free PMC article. Review.
-
"Natural" is not synonymous with "Safe": Toxicity of natural products alone and in combination with pharmaceutical agents.Regul Toxicol Pharmacol. 2020 Jun;113:104642. doi: 10.1016/j.yrtph.2020.104642. Epub 2020 Mar 18. Regul Toxicol Pharmacol. 2020. PMID: 32197968 Free PMC article. Review.
-
Refined Prediction of Pharmacokinetic Kratom-Drug Interactions: Time-Dependent Inhibition Considerations.J Pharmacol Exp Ther. 2021 Jan;376(1):64-73. doi: 10.1124/jpet.120.000270. Epub 2020 Oct 22. J Pharmacol Exp Ther. 2021. PMID: 33093187 Free PMC article.
References
-
- Balentine DA, Wiseman SA, Bouwens LC. (1997) The chemistry of tea flavonoids. Crit Rev Food Sci Nutr 37:693–704. - PubMed
-
- Cheng Y, Prusoff WH. (1973) Relationship between the inhibition constant (KI) and the concentration of inhibitor which causes 50 per cent inhibition (I50) of an enzymatic reaction. Biochem Pharmacol 22:3099–3108. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

