CaV 3.2 drives sustained burst-firing, which is critical for absence seizure propagation in reticular thalamic neurons
- PMID: 29468672
- PMCID: PMC5900875
- DOI: 10.1111/epi.14018
CaV 3.2 drives sustained burst-firing, which is critical for absence seizure propagation in reticular thalamic neurons
Abstract
Objective: Genetic alterations have been identified in the CACNA1H gene, encoding the CaV 3.2 T-type calcium channel in patients with absence epilepsy, yet the precise mechanisms relating to seizure propagation and spike-wave-discharge (SWD) pacemaking remain unknown. Neurons of the thalamic reticular nucleus (TRN) express high levels of CaV 3.2 calcium channels, and we investigated whether a gain-of-function mutation in the Cacna1h gene in Genetic Absence Epilepsy Rats from Strasbourg (GAERS) contributes to seizure propagation and pacemaking in the TRN.
Methods: Pathophysiological contributions of CaV 3.2 calcium channels to burst firing and absence seizures were assessed in vitro using acute brain slice electrophysiology and quantitative real-time polymerase chain reaction (PCR) and in vivo using free-moving electrocorticography recordings.
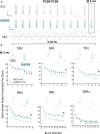
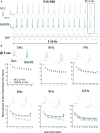
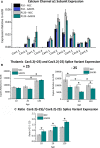
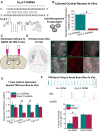
Results: TRN neurons from GAERS display sustained oscillatory burst-firing that is both age- and frequency-dependent, occurring only in the frequencies overlapping with GAERS SWDs and correlating with the expression of a CaV 3.2 mutation-sensitive splice variant. In vivo knock-down of CaV 3.2 using direct thalamic injection of lipid nanoparticles containing CaV 3.2 dicer small interfering (Dsi) RNA normalized TRN burst-firing, and in free-moving GAERS significantly shortened seizures.
Significance: This supports a role for TRN CaV 3.2 T-type channels in propagating thalamocortical network seizures and setting the pacemaking frequency of SWDs.
Keywords: T-type calcium channel; absence epilepsy; low threshold spike; thalamocortical.
© 2018 The Authors. Epilepsia published by Wiley Periodicals, Inc. on behalf of International League Against Epilepsy.
Figures

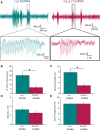
References
-
- Danober L, Deransart C, Depaulis A, et al. Pathophysiological mechanisms of genetic absence epilepsy in the rat. Prog Neurobiol. 1998;55:27–57. - PubMed
-
- Contreras D. The role of T‐channels in the generation of thalamocortical rhythms. CNS Neurol Disord Drug Targets. 2006;5:571–85. - PubMed
-
- Crunelli V, Leresche N. Childhood absence epilepsy: genes, channels, neurons and networks. Nat Rev Neurosci. 2002;3:371–82. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases

