Autosomal recessive Noonan syndrome associated with biallelic LZTR1 variants
- PMID: 29469822
- PMCID: PMC6105555
- DOI: 10.1038/gim.2017.249
Autosomal recessive Noonan syndrome associated with biallelic LZTR1 variants
Abstract
Purpose: To characterize the molecular genetics of autosomal recessive Noonan syndrome.
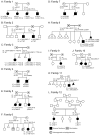
Methods: Families underwent phenotyping for features of Noonan syndrome in children and their parents. Two multiplex families underwent linkage analysis. Exome, genome, or multigene panel sequencing was used to identify variants. The molecular consequences of observed splice variants were evaluated by reverse-transcription polymerase chain reaction.
Results: Twelve families with a total of 23 affected children with features of Noonan syndrome were evaluated. The phenotypic range included mildly affected patients, but it was lethal in some, with cardiac disease and leukemia. All of the parents were unaffected. Linkage analysis using a recessive model supported a candidate region in chromosome 22q11, which includes LZTR1, previously shown to harbor mutations in patients with Noonan syndrome inherited in a dominant pattern. Sequencing analyses of 21 live-born patients and a stillbirth identified biallelic pathogenic variants in LZTR1, including putative loss-of-function, missense, and canonical and noncanonical splicing variants in the affected children, with heterozygous, clinically unaffected parents and heterozygous or normal genotypes in unaffected siblings.
Conclusion: These clinical and genetic data confirm the existence of a form of Noonan syndrome that is inherited in an autosomal recessive pattern and identify biallelic mutations in LZTR1.
Keywords: Noonan syndrome; autosomal recessive inheritance; cardiomyopathy; leukemia; multiple congenital anomalies.
Figures



Comment in
-
Noonan syndrome associated with growth hormone deficiency with biallelic LZTR1 variants.Genet Med. 2019 Jan;21(1):260. doi: 10.1038/s41436-018-0041-5. Epub 2018 Jun 30. Genet Med. 2019. PMID: 29959388 No abstract available.
-
Response to Nakaguma et al.Genet Med. 2019 Jan;21(1):261. doi: 10.1038/s41436-018-0042-4. Epub 2018 Jun 30. Genet Med. 2019. PMID: 29959389 No abstract available.
References
-
- Allanson JE. Objective studies of the face of Noonan, Cardio-facio-cutaneous, and Costello syndromes: A comparison of three disorders of the Ras/MAPK signaling pathway. Am J Med Genet A. 2016;170:2570–2577. - PubMed
-
- Colquitt JL, Noonan JA. Cardiac findings in Noonan syndrome on long-term follow-up. Congenit Heart Dis. 2014;9:144–150. - PubMed
-
- Allanson JE, Roberts AE. Noonan Syndrome. In: Pagon RA, Adam MP, Ardinger HH, et al., editors. GeneReviews(R) Seattle (WA): 1993.
-
- Aoki Y, Niihori T, Inoue S, Matsubara Y. Recent advances in RASopathies. J Hum Genet. 2016;61:33–39. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
- ZIA HG200388/ImNIH/Intramural NIH HHS/United States
- DH_/Department of Health/United Kingdom
- U01 HG007709/HG/NHGRI NIH HHS/United States
- ZIA HG200328-11/HG/NHGRI NIH HHS/United States
- Z01 HG200328/ImNIH/Intramural NIH HHS/United States
- 090532/Z/09/Z/WT_/Wellcome Trust/United Kingdom
- P01 HD070454/HD/NICHD NIH HHS/United States
- ZIA HG200388-04/HG/NHGRI NIH HHS/United States
- ZIA HG200388-03/HG/NHGRI NIH HHS/United States
- ZIA HG200328/ImNIH/Intramural NIH HHS/United States
- WT_/Wellcome Trust/United Kingdom
- ZIA HG200328-12/HG/NHGRI NIH HHS/United States
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases

