The Use of Ex Vivo Organ Cultures in Tick-Borne Virus Research
- PMID: 29473735
- PMCID: PMC6350930
- DOI: 10.1021/acsinfecdis.7b00274
The Use of Ex Vivo Organ Cultures in Tick-Borne Virus Research
Abstract
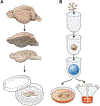
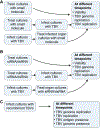
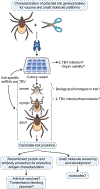
Each year there are more than 15 000 cases of human disease caused by infections with tick-borne viruses (TBVs). These illnesses occur worldwide and can range from very mild illness to severe encephalitis and hemorrhagic fever. Although TBVs are currently identified as neglected vector-borne pathogens and receive less attention than mosquito-borne viruses, TBVs are expanding into new regions, and infection rates are increasing. Furthermore, effective vaccines, diagnostic tools, and other countermeasures are limited. The application of contemporary technologies to TBV infections presents an excellent opportunity to develop improved, effective countermeasures. Experimental tick and mammal models of infection can be used to characterize determinants of infection, transmission, and virulence and to test candidate countermeasures. The use of ex vivo tick cultures in TBV research provides a unique way to look at infection in specific tick organs. Mammal ex vivo organ slice and, more recently, organoid cultures are additional models that can be used to elucidate direct tissue-specific responses to infection. These ex vivo model systems are convenient for testing methods involving transcript knockdown and small molecules under tightly controlled conditions. They can also be combined with in vitro and in vivo studies to tease out possible host factors and potential vaccine or therapeutic candidates. In this brief perspective, we describe how ex vivo cultures can be combined with modern technologies to advance research on TBV infections.
Keywords: flavivirus; organ; therapeutic; tick; vaccine; virus.
Conflict of interest statement
Conflict of Interest
The authors declare no financial competing interests or conflict of interests.
Figures

References
-
- Labuda M; Nuttall PA, Tick-borne viruses. Parasitology 2004, 129 Suppl, S221–245. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

