Cancer vaccine formulation dictates synergy with CTLA-4 and PD-L1 checkpoint blockade therapy
- PMID: 29480817
- PMCID: PMC5873868
- DOI: 10.1172/JCI93303
Cancer vaccine formulation dictates synergy with CTLA-4 and PD-L1 checkpoint blockade therapy
Abstract
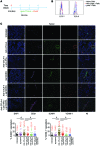
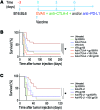
Anticancer vaccination is a promising approach to increase the efficacy of cytotoxic T lymphocyte-associated protein 4 (CTLA-4) and programmed death ligand 1 (PD-L1) checkpoint blockade therapies. However, the landmark FDA registration trial for anti-CTLA-4 therapy (ipilimumab) revealed a complete lack of benefit of adding vaccination with gp100 peptide formulated in incomplete Freund's adjuvant (IFA). Here, using a mouse model of melanoma, we found that gp100 vaccination induced gp100-specific effector T cells (Teffs), which dominantly forced trafficking of anti-CTLA-4-induced, non-gp100-specific Teffs away from the tumor, reducing tumor control. The inflamed vaccination site subsequently also sequestered and destroyed anti-CTLA-4-induced Teffs with specificities for tumor antigens other than gp100, reducing the antitumor efficacy of anti-CTLA-4 therapy. Mechanistically, Teffs at the vaccination site recruited inflammatory monocytes, which in turn attracted additional Teffs in a vicious cycle mediated by IFN-γ, CXCR3, ICAM-1, and CCL2, dependent on IFA formulation. In contrast, nonpersistent vaccine formulations based on dendritic cells, viral vectors, or water-soluble peptides potently synergized with checkpoint blockade of both CTLA-4 and PD-L1 and induced complete tumor regression, including in settings of primary resistance to dual checkpoint blockade. We conclude that cancer vaccine formulation can dominantly determine synergy, or lack thereof, with CTLA-4 and PD-L1 checkpoint blockade therapy for cancer.
Keywords: Cancer immunotherapy; Immunology.
Conflict of interest statement
Figures








References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Research Materials
Miscellaneous

