Metabolic and molecular framework for the enhancement of endurance by intermittent food deprivation
- PMID: 29485903
- PMCID: PMC5998977
- DOI: 10.1096/fj.201701378RR
Metabolic and molecular framework for the enhancement of endurance by intermittent food deprivation
Abstract
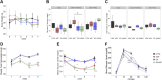
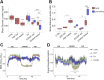
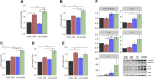
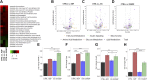
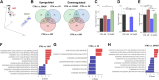
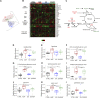
Evolutionary considerations suggest that the body has been optimized to perform at a high level in the food-deprived state when fatty acids and their ketone metabolites are a major fuel source for muscle cells. Because controlled food deprivation in laboratory animals and intermittent energy restriction in humans is a potent physiologic stimulus for ketosis, we designed a study to determine the impact of intermittent food deprivation during endurance training on performance and to elucidate the underlying cellular and molecular mechanisms. Male mice were randomly assigned to either ad libitum feeding or alternate-day food deprivation (ADF) groups, and half of the mice in each diet group were trained daily on a treadmill for 1 mo. A run to exhaustion endurance test performed at the end of the training period revealed superior performance in the mice maintained on ADF during training compared to mice fed ad libitum during training. Maximal O2 consumption was increased similarly by treadmill training in mice on ADF or ad libitum diets, whereas respiratory exchange ratio was reduced in ADF mice on food-deprivation days and during running. Analyses of gene expression in liver and soleus tissues, and metabolomics analysis of blood suggest that the metabolic switch invoked by ADF and potentiated by exercise strongly modulates molecular pathways involved in mitochondrial biogenesis, metabolism, and cellular plasticity. Our findings demonstrate that ADF engages metabolic and cellular signaling pathways that result in increased metabolic efficiency and endurance capacity.-Marosi, K., Moehl, K., Navas-Enamorado, I., Mitchell, S. J., Zhang, Y., Lehrmann, E., Aon, M. A., Cortassa, S., Becker, K. G., Mattson, M. P. Metabolic and molecular framework for the enhancement of endurance by intermittent food deprivation.
Keywords: exercise; intermittent fasting; ketone; mitochondrial biogenesis; muscle.
Conflict of interest statement
This research was supported by the Intramural Research Program of the U.S. National Institutes of Health, National Institute of Aging. The authors declare no conflicts of interest.
Figures


References
-
- Hawkes K., O’Connell J. F., Jones N. G. (1991) Hunting income patterns among the Hadza: big game, common goods, foraging goals and the evolution of the human diet. Philos. Trans. R. Soc. Lond. B Biol. Sci. 334, 243–250 - PubMed
-
- Stanford C. B., Wallis J., Matama H., Goodall J. (1994) Patterns of predation by chimpanzees on red colobus monkeys in Gombe National Park, 1982–1991. Am. J. Phys. Anthropol. 94, 213–228 - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials

