Caspase-Dependent Apoptosis Induction via Viral Protein ORF4 of Porcine Circovirus 2 Binding to Mitochondrial Adenine Nucleotide Translocase 3
- PMID: 29491154
- PMCID: PMC5923066
- DOI: 10.1128/JVI.00238-18
Caspase-Dependent Apoptosis Induction via Viral Protein ORF4 of Porcine Circovirus 2 Binding to Mitochondrial Adenine Nucleotide Translocase 3
Abstract
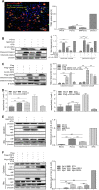
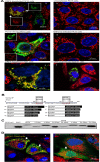
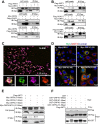
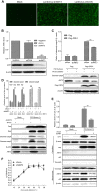
Apoptosis is an essential strategy of host defense responses and is used by viruses to maintain their life cycles. However, the apoptotic signals involved in virus replication are poorly known. In the present study, we report the molecular mechanism of apoptotic induction by the viral protein ORF4, a newly identified viral protein of porcine circovirus type 2 (PCV2). Apoptosis detection revealed not only that the activity of caspase-3 and -9 is increased in PCV2-infected and ORF4-transfected cells but also that cytochrome c release from the mitochondria to the cytosol is upregulated. Subsequently, ORF4 protein colocalization with adenine nucleotide translocase 3 (ANT3) was observed using structured illumination microscopy. Moreover, coimmunoprecipitation and pulldown analyses confirmed that the ORF4 protein interacts directly with mitochondrial ANT3 (mtANT3). Binding domain analysis further confirmed that N-terminal residues 1 to 30 of the ORF4 protein, comprising a mitochondrial targeting signal, are essential for the interaction with ANT3. Knockdown of ANT3 markedly inhibited the apoptotic induction of both ORF4 protein and PCV2, indicating that ANT3 plays an important role in ORF4 protein-induced apoptosis during PCV2 infection. Taken together, these data indicate that the ORF4 protein is a mitochondrial targeting protein that induces apoptosis by interacting with ANT3 through the mitochondrial pathway.IMPORTANCE The porcine circovirus type 2 (PCV2) protein ORF4 is a newly identified viral protein; however, little is known about its functions. Apoptosis is an essential strategy of the host defense response and is used by viruses to maintain their life cycles. In the present study, we report the molecular mechanism of the apoptosis induced by the ORF4 protein. The ORF4 protein contains a mitochondrial targeting signal and is an unstable protein that is degraded by the proteasome-dependent pathway. Viral protein ORF4 triggers caspase-3- and -9-dependent cellular apoptosis in mitochondria by directly binding to ANT3. We conclude that the ORF4 protein is a mitochondrial targeting protein and reveal a mechanism whereby circovirus recruits ANT3 to induce apoptosis.
Keywords: ANT3; ORF4; mitochondrial apoptosis pathway; porcine circovirus; viral protein.
Copyright © 2018 American Society for Microbiology.
Figures
Similar articles
-
The ORF4 protein of porcine circovirus type 2 antagonizes apoptosis by stabilizing the concentration of ferritin heavy chain through physical interaction.J Gen Virol. 2016 Jul;97(7):1636-1646. doi: 10.1099/jgv.0.000472. Epub 2016 Mar 31. J Gen Virol. 2016. PMID: 27030984
-
ORF4-protein deficient PCV2 mutants enhance virus-induced apoptosis and show differential expression of mRNAs in vitro.Virus Res. 2014 Apr;183:56-62. doi: 10.1016/j.virusres.2014.01.024. Epub 2014 Feb 3. Virus Res. 2014. PMID: 24503223
-
Identification and functional analysis of the novel ORF4 protein encoded by porcine circovirus type 2.J Virol. 2013 Feb;87(3):1420-9. doi: 10.1128/JVI.01443-12. Epub 2012 Nov 14. J Virol. 2013. PMID: 23152517 Free PMC article.
-
Porcine circovirus type 2 (PCV2): pathogenesis and interaction with the immune system.Annu Rev Anim Biosci. 2013 Jan;1:43-64. doi: 10.1146/annurev-animal-031412-103720. Epub 2013 Jan 3. Annu Rev Anim Biosci. 2013. PMID: 25387012 Review.
-
Interactions of porcine circovirus 2 with its hosts.Virus Genes. 2016 Aug;52(4):437-44. doi: 10.1007/s11262-016-1326-x. Epub 2016 Mar 25. Virus Genes. 2016. PMID: 27016220 Review.
Cited by
-
Genome-wide analysis of long non-coding RNA expression profile in porcine circovirus 2-infected intestinal porcine epithelial cell line by RNA sequencing.PeerJ. 2019 Mar 6;7:e6577. doi: 10.7717/peerj.6577. eCollection 2019. PeerJ. 2019. PMID: 30863688 Free PMC article.
-
The combined usage of Matrine and Osthole inhibited endoplasmic reticulum apoptosis induced by PCV2.BMC Microbiol. 2020 Oct 12;20(1):303. doi: 10.1186/s12866-020-01986-2. BMC Microbiol. 2020. PMID: 33046006 Free PMC article.
-
Cross-species transmission resulted in the emergence and establishment of circovirus in pig.Infect Genet Evol. 2019 Nov;75:103973. doi: 10.1016/j.meegid.2019.103973. Epub 2019 Jul 19. Infect Genet Evol. 2019. PMID: 31330311 Free PMC article. No abstract available.
-
Functional mutations of SARS-CoV-2: implications to viral transmission, pathogenicity and immune escape.Chin Med J (Engl). 2022 May 20;135(10):1213-1222. doi: 10.1097/CM9.0000000000002158. Chin Med J (Engl). 2022. PMID: 35788093 Free PMC article.
-
Detection and genotyping of Porcine circovirus 2 (PCV-2) and detection of Porcine circovirus 3 (PCV-3) in sera from fattening pigs of different European countries.Transbound Emerg Dis. 2020 Nov;67(6):2521-2531. doi: 10.1111/tbed.13596. Epub 2020 May 14. Transbound Emerg Dis. 2020. PMID: 32356364 Free PMC article.
References
-
- Biagini P, Hino BMS, Kakkola L, Mankertz A, Niel C, Okamoto RSH, Teo CG, Todd D. 2012. Circoviridae, p 343–349. In Virus taxonomy: ninth report of the International Committee on Taxonomy of Viruses. Academic Press, London, United Kingdom.
-
- Tischer I, Rasch R, Tochtermann G. 1974. Characterization of papovavirus- and picornavirus-like particles in permanent pig kidney cell lines. Zentralbl Bakteriol Orig A 226:153–167. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

