Kindlin-2 regulates mesenchymal stem cell differentiation through control of YAP1/TAZ
- PMID: 29496737
- PMCID: PMC5881491
- DOI: 10.1083/jcb.201612177
Kindlin-2 regulates mesenchymal stem cell differentiation through control of YAP1/TAZ
Abstract
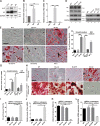
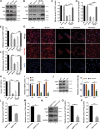
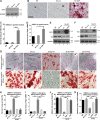
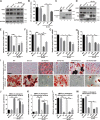
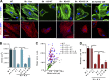
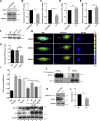
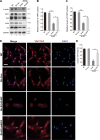
Precise control of mesenchymal stem cell (MSC) differentiation is critical for tissue development and regeneration. We show here that kindlin-2 is a key determinant of MSC fate decision. Depletion of kindlin-2 in MSCs is sufficient to induce adipogenesis and inhibit osteogenesis in vitro and in vivo. Mechanistically, kindlin-2 regulates MSC differentiation through controlling YAP1/TAZ at both the transcript and protein levels. Kindlin-2 physically associates with myosin light-chain kinase in response to mechanical cues of cell microenvironment and intracellular signaling events and promotes myosin light-chain phosphorylation. Loss of kindlin-2 inhibits RhoA activation and reduces myosin light-chain phosphorylation, stress fiber formation, and focal adhesion assembly, resulting in increased Ser127 phosphorylation, nuclear exclusion, and ubiquitin ligase atrophin-1 interacting protein 4-mediated degradation of YAP1/TAZ. Our findings reveal a novel kindlin-2 signaling axis that senses the mechanical cues of cell microenvironment and controls MSC fate decision, and they suggest a new strategy to regulate MSC differentiation, tissue repair, and regeneration.
© 2018 Guo et al.
Figures



Comment in
-
Mechanotransduction: Kindlin' the fate of mesenchymal stem cells.Nat Rev Mol Cell Biol. 2018 May;19(5):278-279. doi: 10.1038/nrm.2018.21. Epub 2018 Mar 21. Nat Rev Mol Cell Biol. 2018. PMID: 29559736 No abstract available.
Similar articles
-
CCM3 is a gatekeeper in focal adhesions regulating mechanotransduction and YAP/TAZ signalling.Nat Cell Biol. 2021 Jul;23(7):758-770. doi: 10.1038/s41556-021-00702-0. Epub 2021 Jul 5. Nat Cell Biol. 2021. Retraction in: Nat Cell Biol. 2024 Oct;26(10):1817. doi: 10.1038/s41556-024-01531-7. PMID: 34226698 Retracted.
-
Type I collagen deposition via osteoinduction ameliorates YAP/TAZ activity in 3D floating culture clumps of mesenchymal stem cell/extracellular matrix complexes.Stem Cell Res Ther. 2018 Dec 7;9(1):342. doi: 10.1186/s13287-018-1085-9. Stem Cell Res Ther. 2018. PMID: 30526677 Free PMC article.
-
A cell-based screening for TAZ activators identifies ethacridine, a widely used antiseptic and abortifacient, as a compound that promotes dephosphorylation of TAZ and inhibits adipogenesis in C3H10T1/2 cells.J Biochem. 2015 Nov;158(5):413-23. doi: 10.1093/jb/mvv051. Epub 2015 May 14. J Biochem. 2015. PMID: 25979969
-
The role of lamin A/C in mesenchymal stem cell differentiation.J Physiol Biochem. 2019 Feb;75(1):11-18. doi: 10.1007/s13105-019-00661-z. Epub 2019 Jan 31. J Physiol Biochem. 2019. PMID: 30706289 Review.
-
The Hippo Pathway as Drug Targets in Cancer Therapy and Regenerative Medicine.Curr Drug Targets. 2017;18(4):447-454. doi: 10.2174/1389450117666160112115641. Curr Drug Targets. 2017. PMID: 26758663 Review.
Cited by
-
CLP36 promotes p53 deficient sarcoma progression through suppression of atrophin-1 interacting protein-4 (AIP-4)-dependent degradation of YAP1.Theranostics. 2022 Jul 4;12(11):5051-5068. doi: 10.7150/thno.72365. eCollection 2022. Theranostics. 2022. PMID: 35836803 Free PMC article.
-
Mechano-regulation of proline metabolism and cancer progression by kindlin-2.Mol Cell Oncol. 2019 Apr 12;6(3):1596003. doi: 10.1080/23723556.2019.1596003. eCollection 2019. Mol Cell Oncol. 2019. PMID: 31131313 Free PMC article.
-
PINCH-1 promotes Δ1-pyrroline-5-carboxylate synthase expression and contributes to proline metabolic reprogramming in lung adenocarcinoma.Amino Acids. 2021 Dec;53(12):1875-1890. doi: 10.1007/s00726-021-03050-3. Epub 2021 Jul 20. Amino Acids. 2021. PMID: 34283311
-
Kindlin-2 promotes Src-mediated tyrosine phosphorylation of androgen receptor and contributes to breast cancer progression.Cell Death Dis. 2022 May 20;13(5):482. doi: 10.1038/s41419-022-04945-z. Cell Death Dis. 2022. PMID: 35595729 Free PMC article.
-
Proteogenomic Analysis Reveals Proteins Involved in the First Step of Adipogenesis in Human Adipose-Derived Stem Cells.Stem Cells Int. 2021 Dec 16;2021:3168428. doi: 10.1155/2021/3168428. eCollection 2021. Stem Cells Int. 2021. PMID: 34956370 Free PMC article.
References
-
- Bredella M.A., Torriani M., Ghomi R.H., Thomas B.J., Brick D.J., Gerweck A.V., Rosen C.J., Klibanski A., and Miller K.K.. 2011. Vertebral bone marrow fat is positively associated with visceral fat and inversely associated with IGF-1 in obese women. Obesity (Silver Spring). 19:49–53. 10.1038/oby.2010.106 - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials

