Ischemic preconditioning attenuates ischemia/reperfusion-induced kidney injury by activating autophagy via the SGK1 signaling pathway
- PMID: 29497029
- PMCID: PMC5832808
- DOI: 10.1038/s41419-018-0358-7
Ischemic preconditioning attenuates ischemia/reperfusion-induced kidney injury by activating autophagy via the SGK1 signaling pathway
Abstract
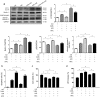
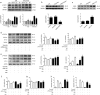
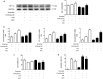
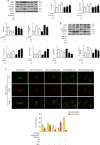
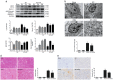
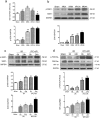
Ischemic preconditioning (IPC) has a strong renoprotective effect during renal ischemia/reperfusion (I/R) injury that is thought to relate to autophagy. However, the role of autophagy during IPC-afforded renoprotection and the precise mechanisms involved are unknown. In this study, an in vitro hypoxia/reoxygenation (H/R) model was established in which oxygen and glucose deprivation (OGD) was applied to renal cells for 15 h followed by reoxygenation under normal conditions for 30 min, 2 h or 6 h; transient OGD and subsequent reoxygenation were implemented before prolonged H/R injury to achieve hypoxic preconditioning (HPC). 3-Methyladenine (3-MA) was used to inhibit autophagy. In a renal I/R injury model, rats were subjected to 40 min of renal ischemia followed by 6 h, 12 h or 24 h of reperfusion. IPC was produced by four cycles of ischemia (8 min each) followed by 5 min of reperfusion prior to sustained ischemia. We found that IPC increased LC3II and Beclin-1 levels and decreased SQSTM/p62 and cleaved caspase-3 levels in a time-dependent manner during renal I/R injury, as well as increased the number of intracellular double-membrane vesicles in injured renal cells. IPC-induced renal protection was efficiently attenuated by pretreatment with 5 mM 3-MA. Pretreatment with IPC also dynamically affected the expression of SGK1/FOXO3a/HIF-1α signaling components. Moreover, knocking down SGK1 expression significantly downregulated phosphorylated-FOXO3a (p-FOXO3a)/FOXO3 and HIF-1α, suppressed LC3II and Beclin-1 levels, increased SQSTM/p62 and cleaved caspase-3 levels, and abolished the protective effect of IPC against I/R-induced renal damage. SGK1 overexpression efficiently increased p-FOXO3a/FOXO3 and HIF-1α levels, promoted the autophagy flux and enhanced the protective effect mediated by HPC. Furthermore, FOXO3a overexpression decreased HIF-1α protein levels, inhibited HIF-1α transcriptional activity and reduced the protective effect of IPC. Our study indicates that IPC can ameliorate renal I/R injury by promoting autophagy through the SGK1 pathway.
Conflict of interest statement
The authors declare that they have no conflict of interest.
Figures
Similar articles
-
Clearance of damaged mitochondria via mitophagy is important to the protective effect of ischemic preconditioning in kidneys.Autophagy. 2019 Dec;15(12):2142-2162. doi: 10.1080/15548627.2019.1615822. Epub 2019 May 22. Autophagy. 2019. PMID: 31066324 Free PMC article.
-
Ischemic Preconditioning Promotes Autophagy and Alleviates Renal Ischemia/Reperfusion Injury.Biomed Res Int. 2018 Jan 22;2018:8353987. doi: 10.1155/2018/8353987. eCollection 2018. Biomed Res Int. 2018. PMID: 29607326 Free PMC article.
-
Ischemic preconditioning improved renal ischemia/reperfusion injury and hyperglycemia.IUBMB Life. 2019 Mar;71(3):321-329. doi: 10.1002/iub.1972. Epub 2018 Nov 27. IUBMB Life. 2019. PMID: 30481400
-
Hypoxic preconditioning in renal ischaemia-reperfusion injury: a review in pre-clinical models.Clin Sci (Lond). 2021 Dec 10;135(23):2607-2618. doi: 10.1042/CS20210615. Clin Sci (Lond). 2021. PMID: 34878507 Review.
-
Is Renal Ischemic Preconditioning an Alternative to Ameliorate the Short- and Long-Term Consequences of Acute Kidney Injury?Int J Mol Sci. 2023 May 6;24(9):8345. doi: 10.3390/ijms24098345. Int J Mol Sci. 2023. PMID: 37176051 Free PMC article. Review.
Cited by
-
Low-dose dexamethasone promotes osteoblast viability by activating autophagy via the SGK1/FOXO3a signaling pathway.Cell Biol Int. 2023 Mar;47(3):669-678. doi: 10.1002/cbin.11971. Epub 2022 Dec 1. Cell Biol Int. 2023. PMID: 36453461 Free PMC article.
-
MeCP2 inhibits cell functionality through FoxO3a and autophagy in endothelial progenitor cells.Aging (Albany NY). 2019 Sep 2;11(17):6714-6733. doi: 10.18632/aging.102183. Epub 2019 Sep 2. Aging (Albany NY). 2019. PMID: 31477637 Free PMC article.
-
The neuroprotective effects of curcumin are associated with the regulation of the reciprocal function between autophagy and HIF-1α in cerebral ischemia-reperfusion injury.Drug Des Devel Ther. 2019 Apr 9;13:1135-1144. doi: 10.2147/DDDT.S194182. eCollection 2019. Drug Des Devel Ther. 2019. PMID: 31040648 Free PMC article.
-
Caffeine Induces Autophagy and Apoptosis in Auditory Hair Cells via the SGK1/HIF-1α Pathway.Front Cell Dev Biol. 2021 Nov 16;9:751012. doi: 10.3389/fcell.2021.751012. eCollection 2021. Front Cell Dev Biol. 2021. PMID: 34869338 Free PMC article.
-
Nephroprotective Role of Chrysophanol in Hypoxia/Reoxygenation-Induced Renal Cell Damage via Apoptosis, ER Stress, and Ferroptosis.Biomedicines. 2021 Sep 21;9(9):1283. doi: 10.3390/biomedicines9091283. Biomedicines. 2021. PMID: 34572468 Free PMC article.
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials

