Minoxidil improves vascular compliance, restores cerebral blood flow, and alters extracellular matrix gene expression in a model of chronic vascular stiffness
- PMID: 29498532
- PMCID: PMC6087770
- DOI: 10.1152/ajpheart.00683.2017
Minoxidil improves vascular compliance, restores cerebral blood flow, and alters extracellular matrix gene expression in a model of chronic vascular stiffness
Abstract
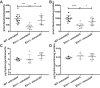
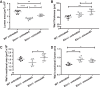
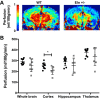
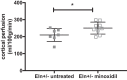
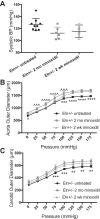
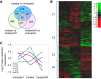
Increased vascular stiffness correlates with a higher risk of cardiovascular complications in aging adults. Elastin (ELN) insufficiency, as observed in patients with Williams-Beuren syndrome or with familial supravalvular aortic stenosis, also increases vascular stiffness and leads to arterial narrowing. We used Eln+/- mice to test the hypothesis that pathologically increased vascular stiffness with concomitant arterial narrowing leads to decreased blood flow to end organs such as the brain. We also hypothesized that drugs that remodel arteries and increase lumen diameter would improve flow. To test these hypotheses, we compared carotid blood flow using ultrasound and cerebral blood flow using MRI-based arterial spin labeling in wild-type (WT) and Eln+/- mice. We then studied how minoxidil, an ATP-sensitive K+ channel opener and vasodilator, affects vessel mechanics, blood flow, and gene expression. Both carotid and cerebral blood flows were lower in Eln+/- mice than in WT mice. Treatment of Eln+/- mice with minoxidil lowered blood pressure and reduced functional arterial stiffness to WT levels. Minoxidil also improved arterial diameter and restored carotid and cerebral blood flows in Eln+/- mice. The beneficial effects persisted for weeks after drug removal. RNA-Seq analysis revealed differential expression of 127 extracellular matrix-related genes among the treatment groups. These results indicate that ELN insufficiency impairs end-organ perfusion, which may contribute to the increased cardiovascular risk. Minoxidil, despite lowering blood pressure, improves end-organ perfusion. Changes in matrix gene expression and persistence of treatment effects after drug withdrawal suggest arterial remodeling. Such remodeling may benefit patients with genetic or age-dependent ELN insufficiency. NEW & NOTEWORTHY Our work with a model of chronic vascular stiffness, the elastin ( Eln)+/- mouse, shows reduced brain perfusion as measured by carotid ultrasound and MRI arterial spin labeling. Vessel caliber, functional stiffness, and blood flow improved with minoxidil. The ATP-sensitive K+ channel opener increased Eln gene expression and altered 126 other matrix-associated genes.
Keywords: ATP-sensitive K+ channel; arterial stiffness; cerebral blood flow; elastin; extracellular matrix; vascular remodeling.
Figures




References
-
- Carta L, Wagenseil JE, Knutsen RH, Mariko B, Faury G, Davis EC, Starcher B, Mecham RP, Ramirez F. Discrete contributions of elastic fiber components to arterial development and mechanical compliance. Arterioscler Thromb Vasc Biol 29: 2083–2089, 2009. doi: 10.1161/ATVBAHA.109.193227. - DOI - PMC - PubMed
-
- Coquand-Gandit M, Jacob MP, Fhayli W, Romero B, Georgieva M, Bouillot S, Estève E, Andrieu JP, Brasseur S, Bouyon S, Garcia-Honduvilla N, Huber P, Buján J, Atanasova M, Faury G. Chronic treatment with minoxidil induces elastic fiber neosynthesis and functional improvement in the aorta of aged mice. Rejuvenation Res 20: 218–230, 2017. doi: 10.1089/rej.2016.1874. - DOI - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

