Redundancy in Anaerobic Digestion Microbiomes during Disturbances by the Antibiotic Monensin
- PMID: 29500266
- PMCID: PMC5930344
- DOI: 10.1128/AEM.02692-17
Redundancy in Anaerobic Digestion Microbiomes during Disturbances by the Antibiotic Monensin
Abstract
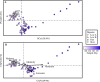
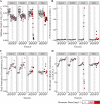
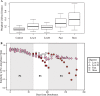
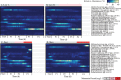
The antibiotic monensin is fed to dairy cows to increase milk production efficiency. A fraction of this monensin is excreted into the cow manure. Previous studies have found that cow manure containing monensin can negatively impact the performance of anaerobic digesters, especially upon first introduction. Few studies have examined whether the anaerobic digester microbiome can adapt to monensin during the operating time. Here, we conducted a long-term time series study of four lab-scale anaerobic digesters fed with cow manure. We examined changes in both the microbiome composition and function of the anaerobic digesters when subjected to the dairy antibiotic monensin. In our digesters, monensin was not rapidly degraded under anaerobic conditions. The two anaerobic digesters that were subjected to manure from monensin feed-dosed cows exhibited relatively small changes in microbiome composition and function due to relatively low monensin concentrations. At higher concentrations of monensin, which we dosed directly to control manure (from dairy cows without monensin), we observed major changes in the microbiome composition and function of two anaerobic digesters. A rapid introduction of monensin to one of these anaerobic digesters led to the impairment of methane production. Conversely, more gradual additions of the same concentrations of monensin to the other anaerobic digester led to the adaptation of the anaerobic digester microbiomes to the relatively high monensin concentrations. A member of the candidate OP11 (Microgenomates) phylum arose in this anaerobic digester and appeared to be redundant with certain Bacteroidetes phylum members, which previously were dominating.IMPORTANCE Monensin is a common antibiotic given to dairy cows in the United States and is partly excreted with dairy manure. An improved understanding of how monensin affects the anaerobic digester microbiome composition and function is important to prevent process failure for farm-based anaerobic digesters. This time series study demonstrates how anaerobic digester microbiomes are inert to low monensin concentrations and can adapt to relatively high monensin concentrations by redundancy in an already existing population. Therefore, our work provides further insight into the importance of microbiome redundancy in maintaining the stability of anaerobic digesters.
Keywords: Bacteroidales; OP11; anaerobic digestion; antibiotic; dairy manure; disturbance; hindgut; microbiome; monensin; redundancy.
Copyright © 2018 Spirito et al.
Figures

References
-
- Fernandez AS, Hashsham SA, Dollhopf SL, Raskin L, Glagoleva O, Dazzo FB, Hickey RF, Criddle CS, Tiedje JM. 2000. Flexible community structure correlates with stable community function in methanogenic bioreactor communities perturbed by glucose. Appl Environ Microbiol 66:4058–4067. doi: 10.1128/AEM.66.9.4058-4067.2000. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

