Vascular Network Formation by Human Microvascular Endothelial Cells in Modular Fibrin Microtissues
- PMID: 29503863
- PMCID: PMC5830175
- DOI: 10.1021/acsbiomaterials.6b00274
Vascular Network Formation by Human Microvascular Endothelial Cells in Modular Fibrin Microtissues
Abstract
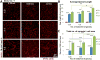
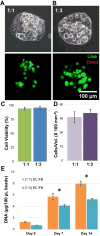
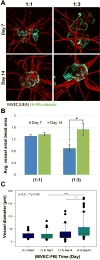
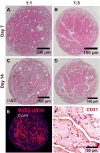
Microvascular endothelial cells (MVEC) are a preferred cell source for autologous revascularization strategies, since they can be harvested and propagated from small tissue biopsies. Biomaterials-based strategies for therapeutic delivery of cells are aimed at tailoring the cellular microenvironment to enhance the delivery, engraftment, and tissue-specific function of transplanted cells. In the present study, we investigated a modular tissue engineering approach to therapeutic revascularization using fibrin-based microtissues containing embedded human MVEC and human fibroblasts (FB). Microtissues were formed using a water-in-oil emulsion process that produced populations of spheroidal tissue modules with a diameter of 100-200 µm. The formation of MVEC sprouts within a fibrin matrix over 7 days in culture was dependent on the presence of FB, with the most robust sprouting occurring at a 1:3 MVEC:FB ratio. Cell viability in microtissues was high (>90%) and significant FB cell proliferation was observed over time in culture. Robust sprouting from microtissues was evident, with larger vessels developing over time and FB acting as pericyte-like cells by enveloping endothelial tubes. These neovessels were shown to form an interconnected vascular plexus over 14 days of culture when microtissues were embedded in a surrounding fibrin hydrogel. Vessel networks exhibited branching and inosculation of sprouts from adjacent microtissues, resulting in MVEC-lined capillaries with hollow lumens. Microtissues maintained in suspension culture aggregated to form larger tissue masses (1-2 mm in diameter) over 7 days. Vessels formed within microtissue aggregates at a 1:1 MVEC:FB ratio were small and diffuse, whereas the 1:3 MVEC:FB ratio produced large and highly interconnected vessels by day 14. This study highlights the utility of human MVEC as a cell source for revascularization strategies, and suggests that the ratio of endothelial to support cells can be used to tailor vessel characteristics. The modular microtissue format may allow minimally invasive delivery of populations of prevascularized microtissues for therapeutic applications.
Keywords: Modular tissue engineering; fibrin; fibrinogen; injectable scaffolds; microtissues; microvascular endothelial cells; minimally invasive delivery; vascularization.
Figures
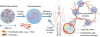
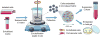



References
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
