Risk of Serious Infection, Opportunistic Infection, and Herpes Zoster among Patients with Psoriasis in the United Kingdom
- PMID: 29505759
- PMCID: PMC6083876
- DOI: 10.1016/j.jid.2018.01.039
Risk of Serious Infection, Opportunistic Infection, and Herpes Zoster among Patients with Psoriasis in the United Kingdom
Abstract
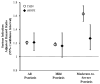
The risk of infection among patients with psoriasis of varying severity in a broadly representative population remains poorly understood. Using The Health Improvement Network (THIN), an electronic medical records database representative of the general UK population, we performed a cohort study to determine the risks of serious infection, opportunistic infection, and herpes zoster among patients with versus without psoriasis and according to psoriasis severity. We identified 187,258 patients with mild and 12,442 patients with moderate to severe psoriasis based on treatment patterns. Using Cox proportional hazards regression, the adjusted hazard ratios (95% confidence intervals) for serious infection were 1.18 (1.16-1.21) and 1.63 (1.52-1.75) for the mild and moderate to severe psoriasis groups, respectively. Among a nested cohort of 8,569 psoriasis patients with disease severity classified by body surface area involvement, similar results were obtained with the exception of an attenuated but significantly increased risk of serious infection among the moderate to severe psoriasis group (1.27 [1.10-1.47]). Overall, the risks of opportunistic infection and herpes zoster were significantly increased only among the moderate to severe psoriasis group and were associated with immunosuppressive therapy. Our analyses suggest that psoriasis is associated with an increased risk of serious infection, and psoriasis severity is a predictor of serious infection risk.
Copyright © 2018 The Authors. Published by Elsevier Inc. All rights reserved.
Conflict of interest statement
Dr. Takeshita receives a research grant (to the Trustees of the University of Pennsylvania) from Pfizer Inc. for work that is unrelated to this manuscript and received payment for continuing medical education work related to psoriasis that was supported indirectly by Eli Lilly and Novartis. Dr. Ogdie has served as a consultant for Bristol-Myers Squibb, Novartis, Pfizer Inc., and Takeda, receiving honoraria; and is a co-investigator on a research grant (to the Trustees of the University of Pennsylvania) from Pfizer Inc. Dr. Gelfand served as a consultant for Coherus (DSMB), Dermira, Janssen Biologics, Merck (DSMB), Novartis Corp, Regeneron, Sanofi, and Pfizer Inc., receiving honoraria; and received research grants (to the Trustees of the University of Pennsylvania) from Abbvie, Janssen, Novartis Corp, Regeneron, Sanofi, Celgene, and Pfizer Inc.; and received payment for continuing medical education work related to psoriasis that was supported indirectly by Eli Lilly and Abbvie. Dr. Gelfand is a co-patent holder of resiquimod for treatment of cutaneous T cell lymphoma.
Figures
References
-
- Austin PC. Using the Standardized Difference to Compare the Prevalence of a Binary Variable Between Two Groups in Observational Research. Commun Stat Simul Comput. 2009;38:1228–34.
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical