Nitrosative damage during retrovirus infection-induced neuropathic pain
- PMID: 29506535
- PMCID: PMC5836380
- DOI: 10.1186/s12974-018-1107-7
Nitrosative damage during retrovirus infection-induced neuropathic pain
Abstract
Background: Peripheral neuropathy is currently the most common neurological complication in HIV-infected individuals, occurring in 35-50% of patients undergoing combination anti-retroviral therapy. Data have shown that distal symmetric polyneuropathy develops in mice by 6 weeks following infection with the LP-BM5 retrovirus mixture. Previous work from our laboratory has demonstrated that glial cells modulate antiviral T-cell effector responses through the programmed death (PD)-1: PD-L1 pathway, thereby limiting the deleterious consequences of unrestrained neuroinflammation.
Methods: Using the MouseMet electronic von Frey system, we assessed hind-paw mechanical hypersensitivity in LP-BM5-infected wild-type (WT) and PD-1 KO animals. Using multi-color flow cytometry, we quantitatively assessed cellular infiltration and microglial activation. Using real-time RT-PCR, we assessed viral load, expression of IFN-γ, iNOS, and MHC class II. Using western blotting, we measured protein nitrosylation within the lumbar spinal cord (LSC) and dorsal root ganglion (DRG). Histochemical staining was performed to analyze the presence of CD3, ionized calcium binding adaptor molecule (Iba)-1, MHCII, nitrotyrosine, isolectin B4 (IB4) binding, and neurofilament 200 (NF200). Statistical analyses were carried out using graphpad prism.
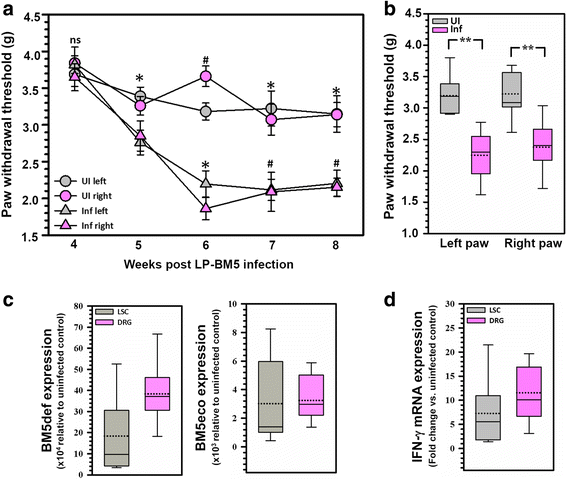
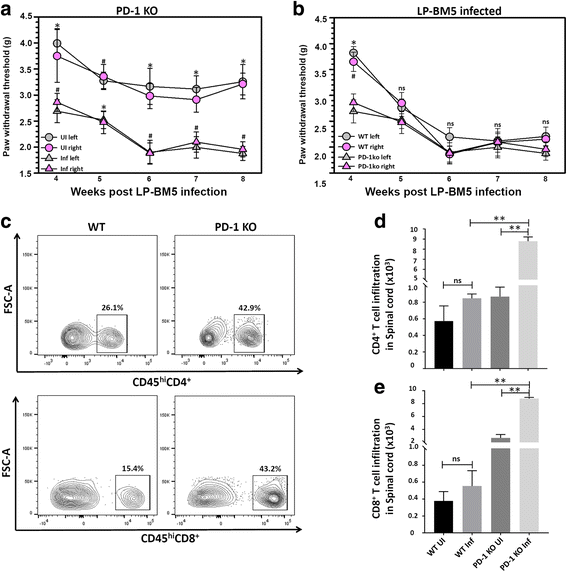
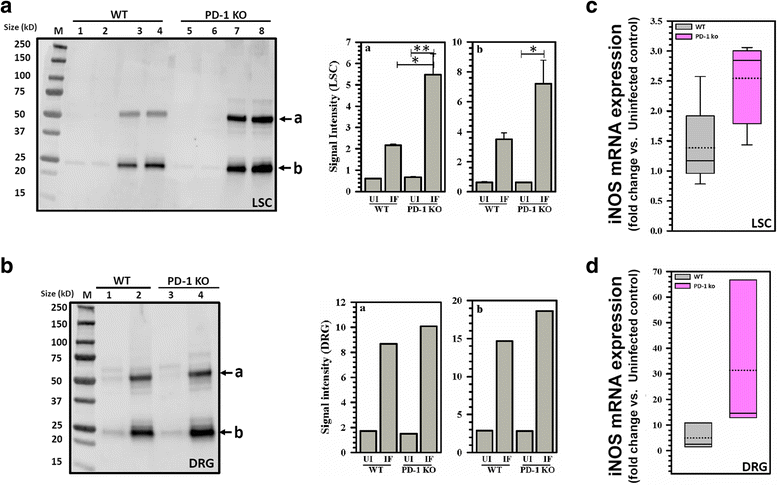
Results: Hind-paw mechanical hypersensitivity observed in LP-BM5-infected animals was associated with significantly increased lymphocyte infiltration into the spinal cord and DRG. We also observed elevated expression of IFN-γ (in LSC and DRG) and MHC II (on resident microglia in LSC). We detected elevated levels of 3-nitrotyrosine within the LSC and DRG of LP-BM5-infected animals, an indicator of nitric oxide (NO)-induced protein damage. Moreover, we observed 3-nitrotyrosine in both small (IB4+) and large (NF200+) DRG sensory neurons. Additionally, infected PD-1 KO animals displayed significantly greater mechanical hypersensitivity than WT or uninfected mice at 4 weeks post-infection (p.i.). Accelerated onset of hind-paw hypersensitivity in PD-1 KO animals was associated with significantly increased infiltration of CD4+ and CD8+ T lymphocytes, macrophages, and microglial activation at early time points. Importantly, we also observed elevated levels of 3-nitrotyrosine and iNOS in infected PD-1 KO animals when compared with WT animals.
Conclusions: Results reported here connect peripheral immune cell infiltration and reactive gliosis with nitrosative damage. These data may help elucidate how retroviral infection-induced neuroinflammatory networks contribute to nerve damage and neuropathic pain.
Keywords: LP-BM5; MAIDS; Neuropathic pain; Nitrosylation; PD-1 KO; Reactive gliosis.
Conflict of interest statement
Ethics approval
This study was carried out in strict accordance with recommendations in the Guide for the Care and Use of Laboratory Animals of the National Institutes of Health. The protocol was approved by the Institutional Animal Care and Use Committee (protocol number: 1709-35110A and breeding protocol number: 1702-34587A) of the University of Minnesota.
Consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Figures






References
-
- Morgello S, Estanislao L, Simpson D, Geraci A, DiRocco A, Gerits P, Ryan E, Yakoushina T, Khan S, Mahboob R, et al. HIV-associated distal sensory polyneuropathy in the era of highly active antiretroviral therapy: the Manhattan HIV brain Bank. Arch Neurol. 2004;61:546–551. doi: 10.1001/archneur.61.4.546. - DOI - PubMed
-
- Ellis RJ, Rosario D, Clifford DB, McArthur JC, Simpson D, Alexander T, Gelman BB, Vaida F, Collier A, Marra CM, et al. Continued high prevalence and adverse clinical impact of human immunodeficiency virus-associated sensory neuropathy in the era of combination antiretroviral therapy: the CHARTER study. Arch Neurol. 2010;67:552–558. doi: 10.1001/archneurol.2010.76. - DOI - PMC - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

