Direct detection and characterization of bioinorganic peroxo moieties in a vanadium complex by 17O solid-state NMR and density functional theory
- PMID: 29506770
- PMCID: PMC6267778
- DOI: 10.1016/j.ssnmr.2018.02.001
Direct detection and characterization of bioinorganic peroxo moieties in a vanadium complex by 17O solid-state NMR and density functional theory
Abstract
Electronic and structural properties of short-lived metal-peroxido complexes, which are key intermediates in many enzymatic reactions, are not fully understood. While detected in various enzymes, their catalytic properties remain elusive because of their transient nature, making them difficult to study spectroscopically. We integrated 17O solid-state NMR and density functional theory (DFT) to directly detect and characterize the peroxido ligand in a bioinorganic V(V) complex mimicking intermediates non-heme vanadium haloperoxidases. 17O chemical shift and quadrupolar tensors, measured by solid-state NMR spectroscopy, probe the electronic structure of the peroxido ligand and its interaction with the metal. DFT analysis reveals the unusually large chemical shift anisotropy arising from the metal orbitals contributing towards the magnetic shielding of the ligand. The results illustrate the power of an integrated approach for studies of oxygen centers in enzyme reaction intermediates.
Keywords: (17)O solid state NMR; Density functional theory calculations; Peroxido ligand; Reactive intermediates.
Copyright © 2018 Elsevier Inc. All rights reserved.
Figures


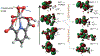
Similar articles
-
Characterization of noninnocent metal complexes using solid-state NMR spectroscopy: o-dioxolene vanadium complexes.Inorg Chem. 2011 Oct 17;50(20):9794-803. doi: 10.1021/ic200046k. Epub 2011 Aug 15. Inorg Chem. 2011. PMID: 21842875 Free PMC article.
-
51V solid-state magic angle spinning NMR spectroscopy and DFT studies of oxovanadium(V) complexes mimicking the active site of vanadium haloperoxidases.Inorg Chem. 2003 Feb 24;42(4):1256-66. doi: 10.1021/ic026141e. Inorg Chem. 2003. PMID: 12588164
-
Investigating the vanadium environments in hydroxylamido V(V) dipicolinate complexes using 51V NMR spectroscopy and density functional theory.Inorg Chem. 2007 Oct 29;46(22):9285-93. doi: 10.1021/ic7012667. Epub 2007 Sep 29. Inorg Chem. 2007. PMID: 17902653 Free PMC article.
-
Manganese-Oxygen Intermediates in O-O Bond Activation and Hydrogen-Atom Transfer Reactions.Acc Chem Res. 2017 Nov 21;50(11):2706-2717. doi: 10.1021/acs.accounts.7b00343. Epub 2017 Oct 24. Acc Chem Res. 2017. PMID: 29064667 Review.
-
Mononuclear metal-O2 complexes bearing macrocyclic N-tetramethylated cyclam ligands.Acc Chem Res. 2012 Aug 21;45(8):1321-30. doi: 10.1021/ar3000019. Epub 2012 May 21. Acc Chem Res. 2012. PMID: 22612523 Free PMC article. Review.
Cited by
-
Oxygen transfer in electrophilic epoxidation probed by 17O NMR: differentiating between oxidants and role of spectator metal oxo.Chem Sci. 2018 Dec 3;10(6):1786-1795. doi: 10.1039/c8sc04868a. eCollection 2019 Feb 14. Chem Sci. 2018. PMID: 30842846 Free PMC article.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

