Structure-based discovery of glycomimetic FmlH ligands as inhibitors of bacterial adhesion during urinary tract infection
- PMID: 29507247
- PMCID: PMC5866590
- DOI: 10.1073/pnas.1720140115
Structure-based discovery of glycomimetic FmlH ligands as inhibitors of bacterial adhesion during urinary tract infection
Abstract
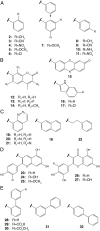
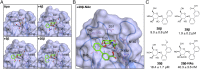
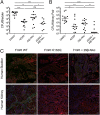
Treatment of bacterial infections is becoming a serious clinical challenge due to the global dissemination of multidrug antibiotic resistance, necessitating the search for alternative treatments to disarm the virulence mechanisms underlying these infections. Uropathogenic Escherichia coli (UPEC) employs multiple chaperone-usher pathway pili tipped with adhesins with diverse receptor specificities to colonize various host tissues and habitats. For example, UPEC F9 pili specifically bind galactose or N-acetylgalactosamine epitopes on the kidney and inflamed bladder. Using X-ray structure-guided methods, virtual screening, and multiplex ELISA arrays, we rationally designed aryl galactosides and N-acetylgalactosaminosides that inhibit the F9 pilus adhesin FmlH. The lead compound, 29β-NAc, is a biphenyl N-acetyl-β-galactosaminoside with a Ki of ∼90 nM, representing a major advancement in potency relative to the characteristically weak nature of most carbohydrate-lectin interactions. 29β-NAc binds tightly to FmlH by engaging the residues Y46 through edge-to-face π-stacking with its A-phenyl ring, R142 in a salt-bridge interaction with its carboxylate group, and K132 through water-mediated hydrogen bonding with its N-acetyl group. Administration of 29β-NAc in a mouse urinary tract infection (UTI) model significantly reduced bladder and kidney bacterial burdens, and coadministration of 29β-NAc and mannoside 4Z269, which targets the type 1 pilus adhesin FimH, resulted in greater elimination of bacteria from the urinary tract than either compound alone. Moreover, FmlH specifically binds healthy human kidney tissue in a 29β-NAc-inhibitable manner, suggesting a key role for F9 pili in human kidney colonization. Thus, these glycoside antagonists of FmlH represent a rational antivirulence strategy for UPEC-mediated UTI treatment.
Keywords: antibiotic-sparing therapeutic; glycomimetics; host–pathogen interactions; structure-based drug design; urinary tract infection.
Copyright © 2018 the Author(s). Published by PNAS.
Conflict of interest statement
Conflict of interest statement: J.W.J. and S.J.H. are inventors on US patent US8937167 B2, which covers the use of mannoside-based FimH ligand antagonists for the treatment of disease. J.W.J., M.E.H., and S.J.H. have ownership interests in Fimbrion Therapeutics and may benefit if the company is successful in marketing mannosides.
Figures


Similar articles
-
Identifying FmlH lectin-binding small molecules for the prevention of Escherichia coli-induced urinary tract infections using hybrid fragment-based design and molecular docking.Comput Biol Med. 2023 Sep;163:107072. doi: 10.1016/j.compbiomed.2023.107072. Epub 2023 Jun 5. Comput Biol Med. 2023. PMID: 37329611 Free PMC article.
-
Antivirulence Isoquinolone Mannosides: Optimization of the Biaryl Aglycone for FimH Lectin Binding Affinity and Efficacy in the Treatment of Chronic UTI.ChemMedChem. 2016 Feb 17;11(4):367-73. doi: 10.1002/cmdc.201600006. Epub 2016 Jan 26. ChemMedChem. 2016. PMID: 26812660 Free PMC article.
-
Inflammation-Induced Adhesin-Receptor Interaction Provides a Fitness Advantage to Uropathogenic E. coli during Chronic Infection.Cell Host Microbe. 2016 Oct 12;20(4):482-492. doi: 10.1016/j.chom.2016.08.013. Epub 2016 Sep 22. Cell Host Microbe. 2016. PMID: 27667696 Free PMC article.
-
Rational design strategies for FimH antagonists: new drugs on the horizon for urinary tract infection and Crohn's disease.Expert Opin Drug Discov. 2017 Jul;12(7):711-731. doi: 10.1080/17460441.2017.1331216. Epub 2017 Jun 2. Expert Opin Drug Discov. 2017. PMID: 28506090 Free PMC article. Review.
-
The Remarkable Biomechanical Properties of the Type 1 Chaperone-Usher Pilus: A Structural and Molecular Perspective.Microbiol Spectr. 2019 Jan;7(1):10.1128/microbiolspec.psib-0010-2018. doi: 10.1128/microbiolspec.PSIB-0010-2018. Microbiol Spectr. 2019. PMID: 30681068 Free PMC article. Review.
Cited by
-
Developments in Mannose-Based Treatments for Uropathogenic Escherichia coli-Induced Urinary Tract Infections.Chembiochem. 2021 Feb 15;22(4):613-629. doi: 10.1002/cbic.202000406. Epub 2020 Nov 2. Chembiochem. 2021. PMID: 32876368 Free PMC article. Review.
-
Structural Basis of Ligand Selectivity by a Bacterial Adhesin Lectin Involved in Multispecies Biofilm Formation.mBio. 2021 Apr 6;12(2):e00130-21. doi: 10.1128/mBio.00130-21. mBio. 2021. PMID: 33824212 Free PMC article.
-
Application of metagenomic next-generation sequencing in the diagnosis of urinary tract infection in patients undergoing cutaneous ureterostomy.Front Cell Infect Microbiol. 2023 Jan 27;13:991011. doi: 10.3389/fcimb.2023.991011. eCollection 2023. Front Cell Infect Microbiol. 2023. PMID: 36779185 Free PMC article.
-
Reaching the End of the Line: Urinary Tract Infections.Microbiol Spectr. 2019 May;7(3):10.1128/microbiolspec.bai-0014-2019. doi: 10.1128/microbiolspec.BAI-0014-2019. Microbiol Spectr. 2019. PMID: 31172909 Free PMC article. Review.
-
Therapeutic Approaches Targeting the Assembly and Function of Chaperone-Usher Pili.EcoSal Plus. 2019 Mar;8(2):10.1128/ecosalplus.ESP-0033-2018. doi: 10.1128/ecosalplus.ESP-0033-2018. EcoSal Plus. 2019. PMID: 30873935 Free PMC article. Review.
References
-
- Foxman B. Epidemiology of urinary tract infections: Incidence, morbidity, and economic costs. Dis Mon. 2003;49:53–70. - PubMed
-
- Griebling TL. Urologic diseases in America project: Trends in resource use for urinary tract infections in women. J Urol. 2005;173:1281–1287. - PubMed
-
- Gupta K, Hooton TM, Roberts PL, Stamm WE. Patient-initiated treatment of uncomplicated recurrent urinary tract infections in young women. Ann Intern Med. 2001;135:9–16. - PubMed
-
- Foxman B. The epidemiology of urinary tract infection. Nat Rev Urol. 2010;7:653–660. - PubMed
-
- Ramakrishnan K, Scheid DC. Diagnosis and management of acute pyelonephritis in adults. Am Fam Physician. 2005;71:933–942. - PubMed
Publication types
MeSH terms
Substances
Associated data
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous

