Continuous-flow retro-Diels-Alder reaction: an efficient method for the preparation of pyrimidinone derivatives
- PMID: 29507637
- PMCID: PMC5815275
- DOI: 10.3762/bjoc.14.20
Continuous-flow retro-Diels-Alder reaction: an efficient method for the preparation of pyrimidinone derivatives
Abstract
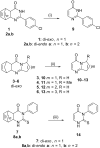
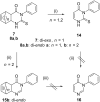
The syntheses of various pyrimidinones as potentially bioactive products by means of the highly controlled continuous-flow retro-Diels-Alder reaction of condensed pyrimidinone derivatives are presented. Noteworthy, the use of this approach allowed us to rapidly screen a selection of conditions and quickly confirm the viability of preparing the desired pyrimidinones in short reaction times. Yields typically higher than those published earlier using conventional batch or microwave processes were achieved.
Keywords: continuous-flow; desulfurisation; norbornene-fused heterocycles; pyrimidinones; retro-Diels–Alder reaction.
Figures

References
-
- Lapkin A A, Plucinski P K. Chemical Reactions; Processes under Flow Conditions. Vol. 5. The Royal Society of. Chemistry; 2010. Engineering Factors for Efficient Flow rocesses in Chemical Industries; pp. 1–43. ((Chapter 1)).
-
- Wegner J, Ceylan S, Kirschning A. Adv Synth Catal. 2012;354:17–57. doi: 10.1002/adsc.201100584. - DOI
-
- Rehm T H, Hofmann C, Reinhard D, Kost H-J, Löb P, Besold M, Welzel K, Barten J, Didenko A, Sevenard D V, et al. React Chem Eng. 2017;2:315–323. doi: 10.1039/c7re00023e. - DOI
-
- Roberge D M, Zimmermann B, Rainone F, Gottsponer M, Eyholzer M, Kockmann N. Org Process Res Dev. 2008;12:905–910. doi: 10.1021/op8001273. - DOI
LinkOut - more resources
Full Text Sources
Other Literature Sources
