Effect of different CT scanners and settings on femoral failure loads calculated by finite element models
- PMID: 29508905
- PMCID: PMC6120464
- DOI: 10.1002/jor.23890
Effect of different CT scanners and settings on femoral failure loads calculated by finite element models
Abstract
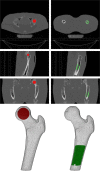
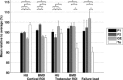
In a multi-center patient study, using different CT scanners, CT-based finite element (FE) models are utilized to calculate failure loads of femora with metastases. Previous studies showed that using different CT scanners can result in different outcomes. This study aims to quantify the effects of (i) different CT scanners; (ii) different CT protocols with variations in slice thickness, field of view (FOV), and reconstruction kernel; and (iii) air between calibration phantom and patient, on Hounsfield Units (HU), bone mineral density (BMD), and FE failure load. Six cadaveric femora were scanned on four CT scanners. Scans were made with multiple CT protocols and with or without an air gap between the body model and calibration phantom. HU and calibrated BMD were determined in cortical and trabecular regions of interest. Non-linear isotropic FE models were constructed to calculate failure load. Mean differences between CT scanners varied up to 7% in cortical HU, 6% in trabecular HU, 6% in cortical BMD, 12% in trabecular BMD, and 17% in failure load. Changes in slice thickness and FOV had little effect (≤4%), while reconstruction kernels had a larger effect on HU (16%), BMD (17%), and failure load (9%). Air between the body model and calibration phantom slightly decreased the HU, BMD, and failure loads (≤8%). In conclusion, this study showed that quantitative analysis of CT images acquired with different CT scanners, and particularly reconstruction kernels, can induce relatively large differences in HU, BMD, and failure loads. Additionally, if possible, air artifacts should be avoided. © 2018 Orthopaedic Research Society. © 2018 The Authors. Journal of Orthopaedic Research® Published by Wiley Periodicals, Inc. on behalf of the Orthopaedic Research Society. J Orthop Res.
Keywords: CT protocol; CT scanner; failure load; femur; finite element model.
© 2018 The Authors. Journal of Orthopaedic Research® Published by Wiley Periodicals, Inc. on behalf of the Orthopaedic Research Society.
Figures


Similar articles
-
The effect of variations in CT scan protocol on femoral finite element failure load assessment using phantomless calibration.PLoS One. 2022 Mar 18;17(3):e0265524. doi: 10.1371/journal.pone.0265524. eCollection 2022. PLoS One. 2022. PMID: 35303026 Free PMC article.
-
Calibration with or without phantom for fracture risk prediction in cancer patients with femoral bone metastases using CT-based finite element models.PLoS One. 2019 Jul 30;14(7):e0220564. doi: 10.1371/journal.pone.0220564. eCollection 2019. PLoS One. 2019. PMID: 31361790 Free PMC article.
-
Standardization of bone mineral density and microstructure from high-resolution CT-scans of the spine in a multicenter setting.Z Med Phys. 2025 May 2:S0939-3889(25)00071-6. doi: 10.1016/j.zemedi.2025.04.002. Online ahead of print. Z Med Phys. 2025. PMID: 40318935
-
Quantitative Computed Tomography (QCT) derived Bone Mineral Density (BMD) in finite element studies: a review of the literature.J Exp Orthop. 2016 Dec;3(1):36. doi: 10.1186/s40634-016-0072-2. Epub 2016 Dec 9. J Exp Orthop. 2016. PMID: 27943224 Free PMC article. Review.
-
Hounsfield Unit-Calculated Bone Mineral Density Loss Following Combat-Related Lower Extremity Amputations.J Bone Joint Surg Am. 2023 Nov 15;105(22):1786-1792. doi: 10.2106/JBJS.22.01258. Epub 2023 Aug 15. J Bone Joint Surg Am. 2023. PMID: 37582168 Review.
Cited by
-
Effect of CT imaging on the accuracy of the finite element modelling in bone.Eur Radiol Exp. 2020 Sep 1;4(1):51. doi: 10.1186/s41747-020-00180-3. Eur Radiol Exp. 2020. PMID: 32869123 Free PMC article.
-
Personalizing Revision Tibial Baseplate Position and Stem Trajectory With Custom Implants Using 3D Modeling to Optimize Press-fit Stem Placement.Arthroplast Today. 2022 Oct 12;18:45-51. doi: 10.1016/j.artd.2022.08.011. eCollection 2022 Dec. Arthroplast Today. 2022. PMID: 36267389 Free PMC article.
-
Asynchronous calibration of a CT scanner for bone mineral density estimation: sources of error and correction.JBMR Plus. 2024 Jul 23;8(9):ziae096. doi: 10.1093/jbmrpl/ziae096. eCollection 2024 Sep. JBMR Plus. 2024. PMID: 39183821 Free PMC article.
-
Quantification of the difference in hounsfield units of an electron density phantom between a conventional and standing computed tomography machine.Vet Res Commun. 2025 Jun 17;49(4):228. doi: 10.1007/s11259-025-10789-7. Vet Res Commun. 2025. PMID: 40526177 Free PMC article.
-
A Patient-Specific Fracture Risk Assessment Tool for Femoral Bone Metastases: Using the Bone Strength (BOS) Score in Clinical Practice.Cancers (Basel). 2022 Nov 29;14(23):5904. doi: 10.3390/cancers14235904. Cancers (Basel). 2022. PMID: 36497388 Free PMC article.
References
-
- Van der Linden YM, Dijkstra PD, Kroon HM, et al. 2004. Comparative analysis of risk factors for pathological fracture with femoral metastases. J Bone Joint Surg Br 86:566–573. - PubMed
-
- Van der Linden YM, Kroon HM, Dijkstra SP, et al. 2003. Simple radiographic parameter predicts fracturing in metastatic femoral bone lesions: results from a randomised trial. Radiother Oncol 69:21–31. - PubMed
-
- Derikx LC, van Aken JB, Janssen D, et al. 2012. The assessment of the risk of fracture in femora with metastatic lesions: comparing case‐specific finite element analyses with predictions by clinical experts. J Bone Joint Surg Br 94:1135–1142. - PubMed
-
- Keyak JH, Kaneko TS, Rossi SA, et al. 2005. Predicting the strength of femoral shafts with and without metastatic lesions. Clin Orthop 439:161–170. - PubMed
-
- Tanck E, van Aken JB, van der Linden YM, et al. 2009. Pathological fracture prediction in patients with metastatic lesions can be improved with quantitative computed tomography based computer models. Bone 45:777–783. - PubMed
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

