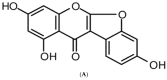
Anti-Inflammatory Effect of Lupinalbin A Isolated from Apios americana on Lipopolysaccharide-Treated RAW264.7 Cells
- PMID: 29509670
- PMCID: PMC6017804
- DOI: 10.3390/molecules23030583
Anti-Inflammatory Effect of Lupinalbin A Isolated from Apios americana on Lipopolysaccharide-Treated RAW264.7 Cells
Abstract
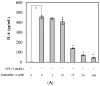
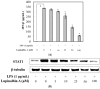
Apios americana, a leguminous plant, is used as food in some countries. Although the biological activities of Apios extract have been reported, there have been no reports about the anti-inflammatory mechanism of lupinalbin A on the RAW264.7 cells. In this study, we investigated the anti-inflammatory effect of A. americana lupinalbin A on lipopolysaccharide (LPS)-treated RAW264.7 cells. Lupinalbin A significantly inhibited nitric oxide production and inducible nitric oxide synthase expression in LPS-treated RAW264.7 cells. The expression of cytokines, including interleukin-6, tumor necrosis factor-α, and chemokine of monocyte chemoattractant protein, was reduced under lupinalbin A exposure in LPS-treated RAW264.7 cells. In addition, lupinalbin A significantly decreased LPS-induced interferon (IFN)-β production and STAT1 protein levels in RAW264.7 cells. Taken together, these results suggest that A. americana lupinalbin A exerts anti-inflammatory effects via the inhibition of pro-inflammatory cytokines and blocking of IFN-β/STAT1 pathway activation.
Keywords: Apios americana; anti-inflammatory effects; lipopolysaccharide; lupinalbin A.
Conflict of interest statement
The authors declare that there are no conflicts of interest.
Figures





Similar articles
-
Anti-Inflammatory Effects of Angelica sinensis (Oliv.) Diels Water Extract on RAW 264.7 Induced with Lipopolysaccharide.Nutrients. 2018 May 21;10(5):647. doi: 10.3390/nu10050647. Nutrients. 2018. PMID: 29883374 Free PMC article.
-
Flavonoids from Apios americana Medikus Leaves Protect RAW264.7 Cells against Inflammation via Inhibition of MAPKs, Akt-mTOR Pathways, and Nfr2 Activation.Oxid Med Cell Longev. 2019 Dec 13;2019:1563024. doi: 10.1155/2019/1563024. eCollection 2019. Oxid Med Cell Longev. 2019. PMID: 31915502 Free PMC article.
-
Apios americana Medikus tuber polysaccharide exerts anti-inflammatory effects by activating autophagy.Int J Biol Macromol. 2019 Jun 1;130:892-902. doi: 10.1016/j.ijbiomac.2019.03.012. Epub 2019 Mar 3. Int J Biol Macromol. 2019. PMID: 30840871
-
Ethyl acetate extract from Asparagus cochinchinensis exerts anti‑inflammatory effects in LPS‑stimulated RAW264.7 macrophage cells by regulating COX‑2/iNOS, inflammatory cytokine expression, MAP kinase pathways, the cell cycle and anti-oxidant activity.Mol Med Rep. 2017 Apr;15(4):1613-1623. doi: 10.3892/mmr.2017.6166. Epub 2017 Feb 3. Mol Med Rep. 2017. PMID: 28260011 Free PMC article.
-
Licocoumarone isolated from Glycyrrhiza uralensis selectively alters LPS-induced inflammatory responses in RAW 264.7 macrophages.Eur J Pharmacol. 2017 Apr 15;801:46-53. doi: 10.1016/j.ejphar.2017.02.049. Epub 2017 Mar 2. Eur J Pharmacol. 2017. PMID: 28263754
Cited by
-
Atraric Acid Exhibits Anti-Inflammatory Effect in Lipopolysaccharide-Stimulated RAW264.7 Cells and Mouse Models.Int J Mol Sci. 2020 Sep 25;21(19):7070. doi: 10.3390/ijms21197070. Int J Mol Sci. 2020. PMID: 32992840 Free PMC article.
-
The Antioxidant, Anti-Inflammatory and Moisturizing Effects of Camellia oleifera Oil and Its Potential Applications.Molecules. 2024 Apr 19;29(8):1864. doi: 10.3390/molecules29081864. Molecules. 2024. PMID: 38675684 Free PMC article.
-
Anti‑inflammatory effect of Chrysanthemum zawadskii, peppermint, Glycyrrhiza glabra herbal mixture in lipopolysaccharide‑stimulated RAW264.7 macrophages.Mol Med Rep. 2021 Jul;24(1):532. doi: 10.3892/mmr.2021.12171. Epub 2021 May 26. Mol Med Rep. 2021. PMID: 34036392 Free PMC article.
-
Comparative metabolomic profiling of Lupinus albus and L. angustifolius harvest residues: exploring chemical diversity and valorization potential.Front Plant Sci. 2025 Jul 7;16:1617634. doi: 10.3389/fpls.2025.1617634. eCollection 2025. Front Plant Sci. 2025. PMID: 40692666 Free PMC article.
-
Anti-diabetic effect of the lupinalbin A compound isolated from Apios americana: In vitro analysis and molecular docking study.Biomed Rep. 2021 Apr;14(4):39. doi: 10.3892/br.2021.1415. Epub 2021 Feb 26. Biomed Rep. 2021. PMID: 33692902 Free PMC article.
References
-
- Marletta M.A. Nitric oxide synthase structure and mechanism. J. Biol. Chem. 1993;268:12231–12234. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

