Uric acid disrupts hypochlorous acid production and the bactericidal activity of HL-60 cells
- PMID: 29510342
- PMCID: PMC5952876
- DOI: 10.1016/j.redox.2018.02.020
Uric acid disrupts hypochlorous acid production and the bactericidal activity of HL-60 cells
Abstract
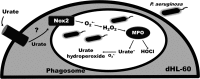
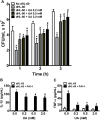
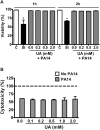
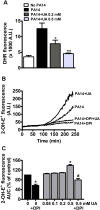
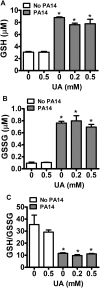
Uric acid is the end product of purine metabolism in humans and is an alternative physiological substrate for myeloperoxidase. Oxidation of uric acid by this enzyme generates uric acid free radical and urate hydroperoxide, a strong oxidant and potentially bactericide agent. In this study, we investigated whether the oxidation of uric acid and production of urate hydroperoxide would affect the killing activity of HL-60 cells differentiated into neutrophil-like cells (dHL-60) against a highly virulent strain (PA14) of the opportunistic pathogen Pseudomonas aeruginosa. While bacterial cell counts decrease due to dHL-60 killing, incubation with uric acid inhibits this activity, also decreasing the release of the inflammatory cytokines interleukin-1β (IL-1β) and tumor necrosis factor-α (TNF- α). In a myeloperoxidase/Cl-/H2O2 cell-free system, uric acid inhibited the production of HOCl and bacterial killing. Fluorescence microscopy showed that uric acid also decreased the levels of HOCl produced by dHL-60 cells, while significantly increased superoxide production. Uric acid did not alter the overall oxidative status of dHL-60 cells as measured by the ratio of reduced (GSH) and oxidized (GSSG) glutathione. Our data show that uric acid impairs the killing activity of dHL-60 cells likely by competing with chloride by myeloperoxidase catalysis, decreasing HOCl production. Despite diminishing HOCl, uric acid probably stimulates the formation of other oxidants, maintaining the overall oxidative status of the cells. Altogether, our results demonstrated that HOCl is, indeed, the main relevant oxidant against bacteria and deviation of myeloperoxidase activity to produce other oxidants hampers dHL-60 killing activity.
Keywords: Hypochlorous acid; Microbicidal; Myeloperoxidase; Pseudomonas aeruginosa; Uric acid; dHL-60.
Copyright © 2018 The Authors. Published by Elsevier B.V. All rights reserved.
Figures



Similar articles
-
Identification of urate hydroperoxide in neutrophils: A novel pro-oxidant generated in inflammatory conditions.Free Radic Biol Med. 2018 Oct;126:177-186. doi: 10.1016/j.freeradbiomed.2018.08.011. Epub 2018 Aug 16. Free Radic Biol Med. 2018. PMID: 30118829
-
Exposure of Pseudomonas aeruginosa to bactericidal hypochlorous acid during neutrophil phagocytosis is compromised in cystic fibrosis.J Biol Chem. 2019 Sep 6;294(36):13502-13514. doi: 10.1074/jbc.RA119.009934. Epub 2019 Jul 24. J Biol Chem. 2019. PMID: 31341024 Free PMC article.
-
Comparative reactivity of the myeloperoxidase-derived oxidants hypochlorous acid and hypothiocyanous acid with human coronary artery endothelial cells.Free Radic Biol Med. 2013 Dec;65:1352-1362. doi: 10.1016/j.freeradbiomed.2013.10.007. Epub 2013 Oct 10. Free Radic Biol Med. 2013. PMID: 24120969
-
Biological reactivity and biomarkers of the neutrophil oxidant, hypochlorous acid.Toxicology. 2002 Dec 27;181-182:223-7. doi: 10.1016/s0300-483x(02)00286-x. Toxicology. 2002. PMID: 12505315 Review.
-
Bacterial Defense Systems against the Neutrophilic Oxidant Hypochlorous Acid.Infect Immun. 2020 Jun 22;88(7):e00964-19. doi: 10.1128/IAI.00964-19. Print 2020 Jun 22. Infect Immun. 2020. PMID: 32152198 Free PMC article. Review.
Cited by
-
Is uric acid a true antioxidant? Identification of uric acid oxidation products and their biological effects.Redox Rep. 2025 Dec;30(1):2498105. doi: 10.1080/13510002.2025.2498105. Epub 2025 May 25. Redox Rep. 2025. PMID: 40415203 Free PMC article. Review.
-
Targeting Myeloperoxidase Ameliorates Gouty Arthritis: A Virtual Screening Success Story.J Med Chem. 2024 Jul 25;67(14):12012-12032. doi: 10.1021/acs.jmedchem.4c00721. Epub 2024 Jul 11. J Med Chem. 2024. PMID: 38991154 Free PMC article.
-
Role of asymptomatic hyperuricemia in the progression of chronic kidney disease and cardiovascular disease.Korean J Intern Med. 2021 Nov;36(6):1281-1293. doi: 10.3904/kjim.2020.340. Epub 2021 Jul 8. Korean J Intern Med. 2021. PMID: 33045808 Free PMC article. Review.
-
Combination of gene/protein and metabolite multiomics to reveal biomarkers of nickel ion cytotoxicity and the underlying mechanism.Regen Biomater. 2024 Jun 29;11:rbae079. doi: 10.1093/rb/rbae079. eCollection 2024. Regen Biomater. 2024. PMID: 39022125 Free PMC article.
-
Hypochlorous Acid: From Innate Immune Factor and Environmental Toxicant to Chemopreventive Agent Targeting Solar UV-Induced Skin Cancer.Front Oncol. 2022 Apr 29;12:887220. doi: 10.3389/fonc.2022.887220. eCollection 2022. Front Oncol. 2022. PMID: 35574306 Free PMC article. Review.
References
-
- Winterbourn C.C., Kettle A.J. Redox reactions and microbial killing in the neutrophil phagosome. Antioxid. Redox Signal. 2013;18:642–660. - PubMed
-
- Winterbourn C.C., Kettle A.J., Hampton M.B. Reactive oxygen species and neutrophil function. Annu. Rev. Biochem. 2016;85:765–792. - PubMed
-
- Ginsburg I., Kohen R. Cell damage in inflammatory and infectious sites might involve a coordinated "cross-talk" among oxidants, microbial haemolysins and ampiphiles, cationic proteins, phospholipases, fatty acids, proteinases and cytokines (an overview) Free Radic. Res. 1995;22:489–517. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

