Ceramide-Protein Interactions Modulate Ceramide-Associated Lipotoxic Cardiomyopathy
- PMID: 29514098
- PMCID: PMC6242704
- DOI: 10.1016/j.celrep.2018.02.034
Ceramide-Protein Interactions Modulate Ceramide-Associated Lipotoxic Cardiomyopathy
Abstract
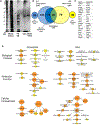
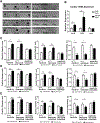
Lipotoxic cardiomyopathy (LCM) is characterized by abnormal myocardial accumulation of lipids, including ceramide; however, the contribution of ceramide to the etiology of LCM is unclear. Here, we investigated the association of ceramide metabolism and ceramide-interacting proteins (CIPs) in LCM in the Drosophila heart model. We find that ceramide feeding or ceramide-elevating genetic manipulations are strongly associated with cardiac dilation and defects in contractility. High ceramide-associated LCM is prevented by inhibiting ceramide synthesis, establishing a robust model of direct ceramide-associated LCM, corroborating previous indirect evidence in mammals. We identified several CIPs from mouse heart and Drosophila extracts, including caspase activator Annexin-X, myosin chaperone Unc-45, and lipogenic enzyme FASN1, and remarkably, their cardiac-specific manipulation can prevent LCM. Collectively, these data suggest that high ceramide-associated lipotoxicity is mediated, in part, through altering caspase activation, sarcomeric maintenance, and lipogenesis, thus providing evidence for conserved mechanisms in LCM pathogenesis in mammals.
Keywords: Annexin; Drosophila; FASN; Unc-45; apoptosis; diabetic cardiac disease; heart; lipogenesis; myriocin; sphingolipids.
Copyright © 2018 The Authors. Published by Elsevier Inc. All rights reserved.
Conflict of interest statement
DECLARATION OF INTERESTS
The authors declare no competing interests.
Figures

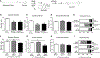



Similar articles
-
Systemic and heart autonomous effects of sphingosine Δ4 desaturase deficiency in lipotoxic cardiac pathophysiology.Dis Model Mech. 2020 Aug 14;13(8):dmm043083. doi: 10.1242/dmm.043083. Dis Model Mech. 2020. PMID: 32641420 Free PMC article.
-
The UNC-45 chaperone is critical for establishing myosin-based myofibrillar organization and cardiac contractility in the Drosophila heart model.PLoS One. 2011;6(7):e22579. doi: 10.1371/journal.pone.0022579. Epub 2011 Jul 25. PLoS One. 2011. PMID: 21799905 Free PMC article.
-
Heart sphingolipids in health and disease.Adv Exp Med Biol. 2011;721:41-56. doi: 10.1007/978-1-4614-0650-1_3. Adv Exp Med Biol. 2011. PMID: 21910081 Review.
-
Type 1 diabetic cardiomyopathy in the Akita (Ins2WT/C96Y) mouse model is characterized by lipotoxicity and diastolic dysfunction with preserved systolic function.Am J Physiol Heart Circ Physiol. 2009 Dec;297(6):H2096-108. doi: 10.1152/ajpheart.00452.2009. Epub 2009 Oct 2. Am J Physiol Heart Circ Physiol. 2009. PMID: 19801494
-
Ceramides - Lipotoxic Inducers of Metabolic Disorders.Trends Endocrinol Metab. 2015 Oct;26(10):538-550. doi: 10.1016/j.tem.2015.07.006. Trends Endocrinol Metab. 2015. PMID: 26412155 Review.
Cited by
-
Ceramides in non-communicable diseases: pathways, nutritional modulation, and therapeutic opportunities.J Physiol Biochem. 2025 Aug 19. doi: 10.1007/s13105-025-01116-4. Online ahead of print. J Physiol Biochem. 2025. PMID: 40828428 Review.
-
Propylene Glycol Alleviates Oxidative Stress and Enhances Immunity in Ketotic Cows through Modulating Amino Acid and Lipid Metabolism.Antioxidants (Basel). 2024 Sep 23;13(9):1146. doi: 10.3390/antiox13091146. Antioxidants (Basel). 2024. PMID: 39334805 Free PMC article.
-
Impact of Drosophila Models in the Study and Treatment of Friedreich's Ataxia.Int J Mol Sci. 2018 Jul 7;19(7):1989. doi: 10.3390/ijms19071989. Int J Mol Sci. 2018. PMID: 29986523 Free PMC article. Review.
-
The Role of Ceramides in Diabetes and Cardiovascular Disease Regulation of Ceramides by Adipokines.Front Endocrinol (Lausanne). 2020 Oct 2;11:569250. doi: 10.3389/fendo.2020.569250. eCollection 2020. Front Endocrinol (Lausanne). 2020. PMID: 33133017 Free PMC article. Review.
-
Systemic and heart autonomous effects of sphingosine Δ4 desaturase deficiency in lipotoxic cardiac pathophysiology.Dis Model Mech. 2020 Aug 14;13(8):dmm043083. doi: 10.1242/dmm.043083. Dis Model Mech. 2020. PMID: 32641420 Free PMC article.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Miscellaneous

