Mutual potentiation drives synergy between trimethoprim and sulfamethoxazole
- PMID: 29520101
- PMCID: PMC5843663
- DOI: 10.1038/s41467-018-03447-x
Mutual potentiation drives synergy between trimethoprim and sulfamethoxazole
Abstract
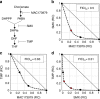
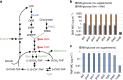
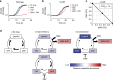
Trimethoprim (TMP)-sulfamethoxazole (SMX) is a widely used synergistic antimicrobial combination to treat a variety of bacterial and certain fungal infections. These drugs act by targeting sequential steps in the biosynthetic pathway for tetrahydrofolate (THF), where SMX inhibits production of the THF precursor dihydropteroate, and TMP inhibits conversion of dihydrofolate (DHF) to THF. Consequently, SMX potentiates TMP by limiting de novo DHF production and this mono-potentiation mechanism is the current explanation for their synergistic action. Here, we demonstrate that this model is insufficient to explain the potent synergy of TMP-SMX. Using genetic and biochemical approaches, we characterize a metabolic feedback loop in which THF is critical for production of the folate precursor dihydropterin pyrophosphate (DHPPP). We reveal that TMP potentiates SMX activity through inhibition of DHPPP synthesis. Our study demonstrates that the TMP-SMX synergy is driven by mutual potentiation of the action of each drug on the other.
Conflict of interest statement
The authors declare no competing interests.
Figures

Similar articles
-
Deletion of nudB Causes Increased Susceptibility to Antifolates in Escherichia coli and Salmonella enterica.Antimicrob Agents Chemother. 2017 Apr 24;61(5):e02378-16. doi: 10.1128/AAC.02378-16. Print 2017 May. Antimicrob Agents Chemother. 2017. PMID: 28223385 Free PMC article.
-
Evaluation of in vitro sensitivity testing methods of detecting sulphamethoxazole trimethoprim synergy. Correlation with mouse protection assay.Arzneimittelforschung. 1980;30(7):1049-51. Arzneimittelforschung. 1980. PMID: 6998489
-
A correlation with type of sulfonamide resistance gene in Escherichia coli and synergy between trimethoprim and sulfamethoxazole.J Basic Microbiol. 1992;32(3):201-7. doi: 10.1002/jobm.3620320309. J Basic Microbiol. 1992. PMID: 1512711
-
The use of trimethoprim and sulfamethoxazole (TMP-SMX) in dermatology.Folia Med Cracov. 2015;55(1):35-41. Folia Med Cracov. 2015. PMID: 26774630 Review.
-
Trimethoprim/sulfamethoxazole for Acinetobacter spp.: A review of current microbiological and clinical evidence.Int J Antimicrob Agents. 2015 Sep;46(3):231-41. doi: 10.1016/j.ijantimicag.2015.04.002. Epub 2015 May 15. Int J Antimicrob Agents. 2015. PMID: 26070662 Review.
Cited by
-
A Strategic Target Rescues Trimethoprim Sensitivity in Escherichia coli.iScience. 2020 Apr 24;23(4):100986. doi: 10.1016/j.isci.2020.100986. Epub 2020 Mar 16. iScience. 2020. PMID: 32240953 Free PMC article.
-
Validation of aminodeoxychorismate synthase and anthranilate synthase as novel targets for bispecific antibiotics inhibiting conserved protein-protein interactions.Appl Environ Microbiol. 2024 May 21;90(5):e0057224. doi: 10.1128/aem.00572-24. Epub 2024 May 3. Appl Environ Microbiol. 2024. PMID: 38700332 Free PMC article.
-
Inhibitor design to target a unique feature in the folate pocket of Staphylococcus aureus dihydrofolate reductase.Eur J Med Chem. 2020 Aug 15;200:112412. doi: 10.1016/j.ejmech.2020.112412. Epub 2020 May 20. Eur J Med Chem. 2020. PMID: 32502861 Free PMC article.
-
Holistic understanding of trimethoprim resistance in Streptococcus pneumoniae using an integrative approach of genome-wide association study, resistance reconstruction, and machine learning.mBio. 2024 Sep 11;15(9):e0136024. doi: 10.1128/mbio.01360-24. Epub 2024 Aug 9. mBio. 2024. PMID: 39120145 Free PMC article.
-
Within-species variability of antibiotic interactions in Gram-negative bacteria.mBio. 2024 Mar 13;15(3):e0019624. doi: 10.1128/mbio.00196-24. Epub 2024 Feb 23. mBio. 2024. PMID: 38391196 Free PMC article.
References
-
- Green, J. M. & Matthews, R. G. Folate biosynthesis, reduction, and polyglutamylation and the interconversion of folate derivatives. EcoSal Plus2,doi:10.1128/ecosalplus.3.6.3.6 (2007). - PubMed
-
- Poe M, Breeze AS, Wu JK, Short CR, Hoogsteen K. Dihydrofolate reductase from trimethoprim-resistant Escherichia coli MB 3746 and MB 3747. Purification, amino acid composition, and some kinetic properties. J. Biol. Chem. 1979;254:1799–1805. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

