Proteomics reveals novel protein associations with early endosomes in an epidermal growth factor-dependent manner
- PMID: 29523688
- PMCID: PMC5912451
- DOI: 10.1074/jbc.RA117.000632
Proteomics reveals novel protein associations with early endosomes in an epidermal growth factor-dependent manner
Abstract
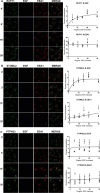
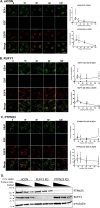
The epidermal growth factor receptor (EGFR) is a receptor tyrosine kinase that is an integral component of proliferative signaling. EGFRs on the cell surface become activated upon EGF binding and have an increased rate of endocytosis. Once in the cytoplasm, the EGF·EGFR complex is trafficked to the lysosome for degradation, and signaling is terminated. During trafficking, the EGFR kinase domain remains active, and the internalized EGFR can continue signaling to downstream effectors. Although effector activity varies based on the EGFR's endocytic location, it is not clear how this occurs. In an effort to identify proteins that uniquely associate with the internalized, liganded EGFR in the early endosome, we developed an early endosome isolation strategy to analyze their protein composition. Post-nuclear supernatant from HeLa cells stimulated with and without EGF were separated on an isotonic 17% Percoll gradient. The gradient was fractionated, and early endosomal fractions were pooled and immunoisolated with an EEA1 mAb. The isolated endosomes were validated by immunoblot using antibodies against organelle-specific marker proteins and transmission EM. These early endosomes were also subjected to LC-MS/MS for proteomic analysis. Five proteins were detected in endosomes in a ligand-dependent manner: EGFR, RUFY1, STOML2, PTPN23, and CCDC51. Knockdown of RUFY1 or PTPN23 by RNAi indicated that both proteins play a role in EGFR trafficking. These experiments indicate that endocytic trafficking of activated EGFR changes the protein composition, membrane trafficking, and signaling potential of the early endosome.
Keywords: endosome; epidermal growth factor receptor (EGFR); membrane trafficking; proteomics; receptor endocytosis.
© 2018 by The American Society for Biochemistry and Molecular Biology, Inc.
Conflict of interest statement
The authors declare that they have no conflicts of interest with the contents of this article
Figures





Similar articles
-
rab7 activity affects epidermal growth factor:epidermal growth factor receptor degradation by regulating endocytic trafficking from the late endosome.J Biol Chem. 2006 Jan 13;281(2):1099-106. doi: 10.1074/jbc.M504175200. Epub 2005 Nov 10. J Biol Chem. 2006. PMID: 16282324
-
Engagement of the small GTPase Rab31 protein and its effector, early endosome antigen 1, is important for trafficking of the ligand-bound epidermal growth factor receptor from the early to the late endosome.J Biol Chem. 2014 May 2;289(18):12375-89. doi: 10.1074/jbc.M114.548321. Epub 2014 Mar 18. J Biol Chem. 2014. PMID: 24644286 Free PMC article.
-
Evidence for efficient phosphorylation of EGFR and rapid endocytosis of phosphorylated EGFR via the early/late endocytic pathway in a gefitinib-sensitive non-small cell lung cancer cell line.Mol Cancer. 2008 May 21;7:42. doi: 10.1186/1476-4598-7-42. Mol Cancer. 2008. PMID: 18492291 Free PMC article.
-
Endocytosis and intracellular trafficking of ErbBs.Exp Cell Res. 2008 Oct 15;314(17):3093-106. doi: 10.1016/j.yexcr.2008.08.013. Epub 2008 Aug 28. Exp Cell Res. 2008. PMID: 18793634 Free PMC article. Review.
-
The physical basis of analog-to-digital signal processing in the EGFR system-Delving into the role of the endoplasmic reticulum.Bioessays. 2024 Sep;46(9):e2400026. doi: 10.1002/bies.202400026. Epub 2024 Jul 11. Bioessays. 2024. PMID: 38991978 Review.
Cited by
-
The RUFYs, a Family of Effector Proteins Involved in Intracellular Trafficking and Cytoskeleton Dynamics.Front Cell Dev Biol. 2020 Aug 11;8:779. doi: 10.3389/fcell.2020.00779. eCollection 2020. Front Cell Dev Biol. 2020. PMID: 32850870 Free PMC article. Review.
-
An optimized fractionation method reveals insulin-induced membrane surface localization of GLUT1 to increase glycolysis in LβT2 cells.Mol Cell Endocrinol. 2025 Jan 1;595:112405. doi: 10.1016/j.mce.2024.112405. Epub 2024 Oct 29. Mol Cell Endocrinol. 2025. PMID: 39481749 Free PMC article.
-
The GPCR adaptor protein Norbin controls the trafficking of C5aR1 and CXCR4 in mouse neutrophils.J Biol Chem. 2024 Dec;300(12):107940. doi: 10.1016/j.jbc.2024.107940. Epub 2024 Oct 28. J Biol Chem. 2024. PMID: 39476960 Free PMC article.
-
RUFY3 and RUFY4 are ARL8 effectors that promote coupling of endolysosomes to dynein-dynactin.Nat Commun. 2022 Mar 21;13(1):1506. doi: 10.1038/s41467-022-28952-y. Nat Commun. 2022. PMID: 35314674 Free PMC article.
-
Influenza A virus exploits transferrin receptor recycling to enter host cells.Proc Natl Acad Sci U S A. 2023 May 23;120(21):e2214936120. doi: 10.1073/pnas.2214936120. Epub 2023 May 16. Proc Natl Acad Sci U S A. 2023. PMID: 37192162 Free PMC article.
References
-
- Nicholson R. I., Gee J. M., and Harper M. E. (2001) EGFR and cancer prognosis. Eur. J. Cancer 37, S9–S15 - PubMed
-
- Garrett T. P., McKern N. M., Lou M., Elleman T. C., Adams T. E., Lovrecz G. O., Zhu H. J., Walker F., Frenkel M. J., Hoyne P. A., Jorissen R. N., Nice E. C., Burgess A. W., and Ward C. W. (2002) Crystal structure of a truncated epidermal growth factor receptor extracellular domain bound to transforming growth factor α. Cell 110, 763–773 10.1016/S0092-8674(02)00940-6 - DOI - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
Miscellaneous

