ABCC1-Exported Sphingosine-1-phosphate, Produced by Sphingosine Kinase 1, Shortens Survival of Mice and Patients with Breast Cancer
- PMID: 29523764
- PMCID: PMC6071310
- DOI: 10.1158/1541-7786.MCR-17-0353
ABCC1-Exported Sphingosine-1-phosphate, Produced by Sphingosine Kinase 1, Shortens Survival of Mice and Patients with Breast Cancer
Abstract
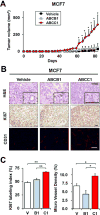
Sphingosine-1-phosphate (S1P), a bioactive sphingolipid mediator, has been implicated in regulation of many processes important for breast cancer progression. Previously, we observed that S1P is exported out of human breast cancer cells by ATP-binding cassette (ABC) transporter ABCC1, but not by ABCB1, both known multidrug resistance proteins that efflux chemotherapeutic agents. However, the pathologic consequences of these events to breast cancer progression and metastasis have not been elucidated. Here, it is demonstrated that high expression of ABCC1, but not ABCB1, is associated with poor prognosis in breast cancer patients. Overexpression of ABCC1, but not ABCB1, in human MCF7 and murine 4T1 breast cancer cells enhanced S1P secretion, proliferation, and migration of breast cancer cells. Implantation of breast cancer cells overexpressing ABCC1, but not ABCB1, into the mammary fat pad markedly enhanced tumor growth, angiogenesis, and lymphangiogenesis with a concomitant increase in lymph node and lung metastases as well as shorter survival of mice. Interestingly, S1P exported via ABCC1 from breast cancer cells upregulated transcription of sphingosine kinase 1 (SPHK1), thus promoting more S1P formation. Finally, patients with breast cancers that express both activated SPHK1 and ABCC1 have significantly shorter disease-free survival. These findings suggest that export of S1P via ABCC1 functions in a malicious feed-forward manner to amplify the S1P axis involved in breast cancer progression and metastasis, which has important implications for prognosis of breast cancer patients and for potential therapeutic targets.Implication: Multidrug resistant transporter ABCC1 and activation of SPHK1 in breast cancer worsen patient's survival by export of S1P to the tumor microenvironment to enhance key processes involved in cancer progression. Mol Cancer Res; 16(6); 1059-70. ©2018 AACR.
©2018 American Association for Cancer Research.
Conflict of interest statement
No potential conflict of interest to disclose.
Figures






Similar articles
-
ATP-binding cassette ABCC1 is involved in the release of sphingosine 1-phosphate from rat uterine leiomyoma ELT3 cells and late pregnant rat myometrium.Cell Signal. 2011 Dec;23(12):1997-2004. doi: 10.1016/j.cellsig.2011.07.010. Epub 2011 Jul 22. Cell Signal. 2011. PMID: 21803151
-
Estradiol induces export of sphingosine 1-phosphate from breast cancer cells via ABCC1 and ABCG2.J Biol Chem. 2010 Apr 2;285(14):10477-86. doi: 10.1074/jbc.M109.064162. Epub 2010 Jan 28. J Biol Chem. 2010. PMID: 20110355 Free PMC article.
-
Sphingosine-1-phosphate produced by sphingosine kinase 1 promotes breast cancer progression by stimulating angiogenesis and lymphangiogenesis.Cancer Res. 2012 Feb 1;72(3):726-35. doi: 10.1158/0008-5472.CAN-11-2167. Cancer Res. 2012. PMID: 22298596 Free PMC article.
-
The role of sphingosine-1-phosphate in breast cancer tumor-induced lymphangiogenesis.Lymphat Res Biol. 2012 Sep;10(3):97-106. doi: 10.1089/lrb.2012.0010. Lymphat Res Biol. 2012. PMID: 22984905 Free PMC article. Review.
-
Sphingosine-1-phosphate and estrogen signaling in breast cancer.Adv Biol Regul. 2016 Jan;60:160-165. doi: 10.1016/j.jbior.2015.09.006. Epub 2015 Oct 19. Adv Biol Regul. 2016. PMID: 26601898 Review.
Cited by
-
Clinical Significance and Potential Mechanisms of ATP Binding Cassette Subfamily C Genes in Hepatocellular Carcinoma.Front Genet. 2022 Mar 7;13:805961. doi: 10.3389/fgene.2022.805961. eCollection 2022. Front Genet. 2022. PMID: 35342392 Free PMC article.
-
Chemoresistance and Metastasis in Breast Cancer Molecular Mechanisms and Novel Clinical Strategies.Front Oncol. 2021 Jul 1;11:658552. doi: 10.3389/fonc.2021.658552. eCollection 2021. Front Oncol. 2021. PMID: 34277408 Free PMC article. Review.
-
Spinster Homologue 2 Expression Correlates With Improved Patient Survival in Hepatocellular Carcinoma Despite Association With Lymph-Angiogenesis.World J Oncol. 2024 Apr;15(2):181-191. doi: 10.14740/wjon1732. Epub 2024 Mar 21. World J Oncol. 2024. PMID: 38545475 Free PMC article.
-
Pan-cancer analysis of ABCC1 as a potential prognostic and immunological biomarker.Transl Oncol. 2024 Mar;41:101882. doi: 10.1016/j.tranon.2024.101882. Epub 2024 Jan 29. Transl Oncol. 2024. PMID: 38290247 Free PMC article.
-
Lipid Metabolism in Breast Cancer: From Basic Research to Clinical Application.Cancers (Basel). 2025 Feb 14;17(4):650. doi: 10.3390/cancers17040650. Cancers (Basel). 2025. PMID: 40002245 Free PMC article. Review.
References
-
- DeSantis CE, Fedewa SA, Goding Sauer A, Kramer JL, Smith RA, Jemal A. Breast cancer statistics, 2015: Convergence of incidence rates between black and white women. CA Cancer J Clin. 2016;66:31–42. - PubMed
-
- Szakacs G, Annereau JP, Lababidi S, Shankavaram U, Arciello A, Bussey KJ, et al. Predicting drug sensitivity and resistance: profiling ABC transporter genes in cancer cells. Cancer cell. 2004;6:129–37. - PubMed
-
- Fletcher JI, Haber M, Henderson MJ, Norris MD. ABC transporters in cancer: more than just drug efflux pumps. Nat Rev Cancer. 2010;10:147–56. - PubMed
-
- Chen Z, Shi T, Zhang L, Zhu P, Deng M, Huang C, et al. Mammalian drug efflux transporters of the ATP binding cassette (ABC) family in multidrug resistance: A review of the past decade. Cancer lett. 2016;370:153–64. - PubMed
-
- Leonessa F, Clarke R. ATP binding cassette transporters and drug resistance in breast cancer. Endocr Relat Cancer. 2003;10:43–73. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

