Translocon Declogger Ste24 Protects against IAPP Oligomer-Induced Proteotoxicity
- PMID: 29526462
- PMCID: PMC5945206
- DOI: 10.1016/j.cell.2018.02.026
Translocon Declogger Ste24 Protects against IAPP Oligomer-Induced Proteotoxicity
Abstract
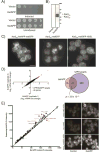
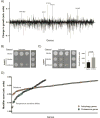
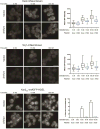
Aggregates of human islet amyloid polypeptide (IAPP) in the pancreas of patients with type 2 diabetes (T2D) are thought to contribute to β cell dysfunction and death. To understand how IAPP harms cells and how this might be overcome, we created a yeast model of IAPP toxicity. Ste24, an evolutionarily conserved protease that was recently reported to degrade peptides stuck within the translocon between the cytoplasm and the endoplasmic reticulum, was the strongest suppressor of IAPP toxicity. By testing variants of the human homolog, ZMPSTE24, with varying activity levels, the rescue of IAPP toxicity proved to be directly proportional to the declogging efficiency. Clinically relevant ZMPSTE24 variants identified in the largest database of exomes sequences derived from T2D patients were characterized using the yeast model, revealing 14 partial loss-of-function variants, which were enriched among diabetes patients over 2-fold. Thus, clogging of the translocon by IAPP oligomers may contribute to β cell failure.
Keywords: IAPP; ZMPSTE24; aggregation; amylin; diabetes; protein folding; proteotoxicity; yeast.
Copyright © 2018 Elsevier Inc. All rights reserved.
Conflict of interest statement
The authors declare no competing interests.
Figures
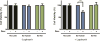



Similar articles
-
The Protease Ste24 Clears Clogged Translocons.Cell. 2016 Jan 14;164(1-2):103-114. doi: 10.1016/j.cell.2015.11.053. Cell. 2016. PMID: 26771486 Free PMC article.
-
IAPP toxicity activates HIF1α/PFKFB3 signaling delaying β-cell loss at the expense of β-cell function.Nat Commun. 2019 Jun 18;10(1):2679. doi: 10.1038/s41467-019-10444-1. Nat Commun. 2019. PMID: 31213603 Free PMC article.
-
Causative factors for formation of toxic islet amyloid polypeptide oligomer in type 2 diabetes mellitus.Clin Interv Aging. 2015 Nov 19;10:1873-9. doi: 10.2147/CIA.S95297. eCollection 2015. Clin Interv Aging. 2015. PMID: 26604727 Free PMC article. Review.
-
Nonhuman IAPP Variants Inhibit Human IAPP Aggregation.Protein Pept Lett. 2021;28(9):963-971. doi: 10.2174/0929866528666210806152706. Protein Pept Lett. 2021. PMID: 34365921 Free PMC article.
-
The β-cell assassin: IAPP cytotoxicity.J Mol Endocrinol. 2017 Oct;59(3):R121-R140. doi: 10.1530/JME-17-0105. Epub 2017 Aug 15. J Mol Endocrinol. 2017. PMID: 28811318 Review.
Cited by
-
Role of β-cell autophagy in β-cell physiology and the development of diabetes.J Diabetes Investig. 2024 Jun;15(6):656-668. doi: 10.1111/jdi.14184. Epub 2024 Mar 12. J Diabetes Investig. 2024. PMID: 38470018 Free PMC article. Review.
-
ZMPSTE24 missense mutations that cause progeroid diseases decrease prelamin A cleavage activity and/or protein stability.Dis Model Mech. 2018 Jul 13;11(7):dmm033670. doi: 10.1242/dmm.033670. Dis Model Mech. 2018. PMID: 29794150 Free PMC article.
-
Heat shock protein Hspa13 regulates endoplasmic reticulum and cytosolic proteostasis through modulation of protein translocation.J Biol Chem. 2022 Dec;298(12):102597. doi: 10.1016/j.jbc.2022.102597. Epub 2022 Oct 14. J Biol Chem. 2022. PMID: 36244454 Free PMC article.
-
Spf1 and Ste24: quality controllers of transmembrane protein topology in the eukaryotic cell.Front Cell Dev Biol. 2023 Aug 3;11:1220441. doi: 10.3389/fcell.2023.1220441. eCollection 2023. Front Cell Dev Biol. 2023. PMID: 37635876 Free PMC article. Review.
-
Prelamin A and ZMPSTE24 in premature and physiological aging.Nucleus. 2023 Dec;14(1):2270345. doi: 10.1080/19491034.2023.2270345. Epub 2023 Oct 26. Nucleus. 2023. PMID: 37885131 Free PMC article. Review.
References
-
- Agarwal AK, Fryns JP, Auchus RJ, Garg A. Zinc metalloproteinase, ZMPSTE24, is mutated in mandibuloacral dysplasia. Hum Mol Genet. 2003;12:1995–2001. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials

