Immune Responses to a Recombinant Glycoprotein E Herpes Zoster Vaccine in Adults Aged 50 Years or Older
- PMID: 29529222
- PMCID: PMC5946839
- DOI: 10.1093/infdis/jiy095
Immune Responses to a Recombinant Glycoprotein E Herpes Zoster Vaccine in Adults Aged 50 Years or Older
Abstract
Background: The herpes zoster subunit vaccine (HZ/su), consisting of varicella-zoster virus glycoprotein E (gE) and AS01B Adjuvant System, was highly efficacious in preventing herpes zoster in the ZOE-50 and ZOE-70 trials. We present immunogenicity results from those trials.
Methods: Participants (ZOE-50: ≥50; ZOE-70: ≥70 years of age) received 2 doses of HZ/su or placebo, 2 months apart. Serum anti-gE antibodies and CD4 T cells expressing ≥2 of 4 activation markers assessed (CD42+) after stimulation with gE-peptides were measured in subcohorts for humoral (n = 3293) and cell-mediated (n = 466) immunogenicity.
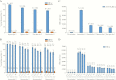
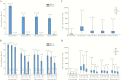
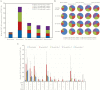
Results: After vaccination, 97.8% of HZ/su and 2.0% of placebo recipients showed a humoral response. Geometric mean anti-gE antibody concentrations increased 39.1-fold and 8.3-fold over baseline in HZ/su recipients at 1 and 36 months post-dose 2, respectively. A gE-specific CD42+ T-cell response was shown in 93.3% of HZ/su and 0% of placebo recipients. Median CD42+ T-cell frequencies increased 24.6-fold (1 month) and 7.9-fold (36 months) over baseline in HZ/su recipients and remained ≥5.6-fold above baseline in all age groups at 36 months. The proportion of CD4 T cells expressing all 4 activation markers increased over time in all age groups.
Conclusions: Most HZ/su recipients developed robust immune responses persisting for 3 years following vaccination.
Clinical trials registration: NCT01165177; NCT01165229.
Figures
References
-
- Levin MJ. Licensed vaccines. Zoster vaccine. In: Orenstein WA, Offit PA, eds. Vaccines. 6th ed London: W.B. Saunders, 2013:969–80.
-
- Lal H, Cunningham AL, Godeaux O et al. ; ZOE-50 Study Group Efficacy of an adjuvanted herpes zoster subunit vaccine in older adults. N Engl J Med 2015; 372:2087–96. - PubMed
-
- Cunningham AL, Lal H, Kovac M et al. ; ZOE-70 Study Group Efficacy of the herpes zoster subunit vaccine in adults 70 years of age or older. N Engl J Med 2016; 375:1019–32. - PubMed
-
- Oxman MN, Levin MJ, Johnson GR et al. ; Shingles Prevention Study Group A vaccine to prevent herpes zoster and postherpetic neuralgia in older adults. N Engl J Med 2005; 352:2271–84. - PubMed
Publication types
MeSH terms
Substances
Associated data
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials

