Kidney stone formers have more renal parenchymal crystals than non-stone formers, particularly in the papilla region
- PMID: 29530009
- PMCID: PMC5848581
- DOI: 10.1186/s12894-018-0331-x
Kidney stone formers have more renal parenchymal crystals than non-stone formers, particularly in the papilla region
Abstract
Background: We investigated the renoprotective ability of healthy people against kidney stone formation. To clarify intratubular crystal kinetics and processing in human kidneys, we performed a quantitative and morphological observation of nephrectomized renal parenchyma tissues.
Methods: Clinical data and pathological samples from 60 patients who underwent radical nephrectomy for renal cancer were collected from June 2004 to June 2010. The patients were retrospectively classified as stone formers (SFs; n = 30, kidney stones detected by preoperative computed tomography) and non-stone formers (NSFs; n = 30, no kidney stone history). The morphology of parenchymal intratubular crystals and kidney stone-related gene and protein expression levels were examined in noncancerous renal sections from both groups.
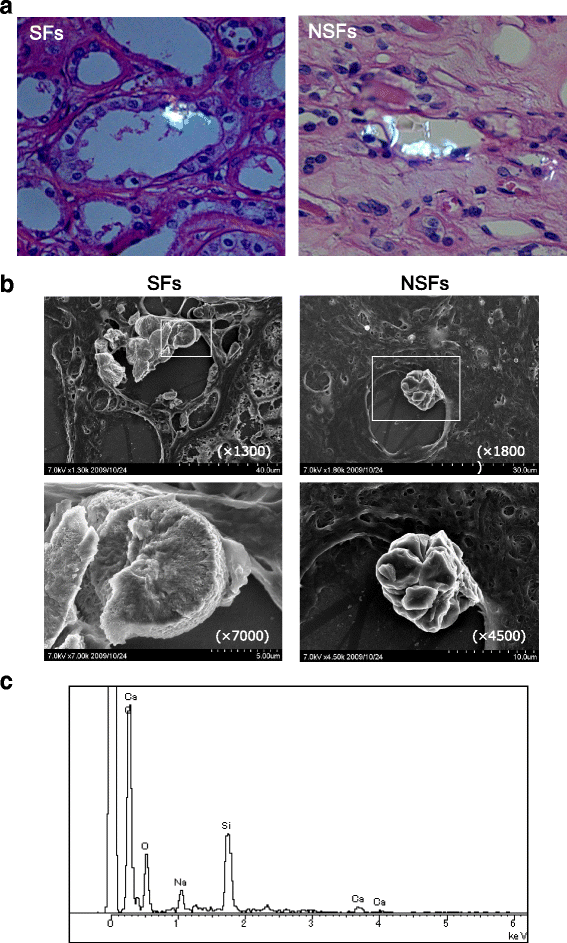
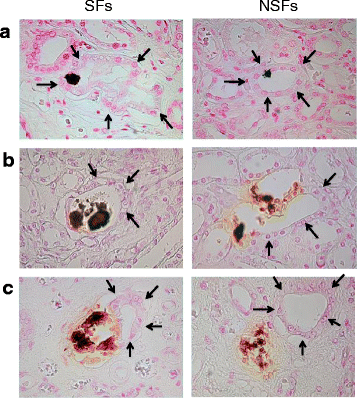
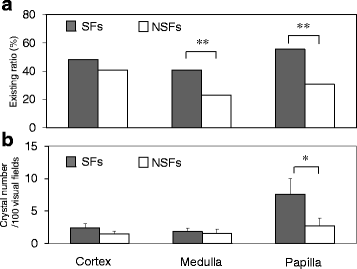
Results: SFs had a higher smoking rate (P = 0.0097); lower red blood cell, hemoglobin, and hematocrit values; and higher urinary red blood cell, white blood cell, and bacterial counts than NSFs. Scanning electron microscopy revealed calcium-containing crystal deposits and crystal attachment to the renal tubular lumen in both groups. Both groups demonstrated crystal transmigration from the tubular lumen to the interstitium. The crystal diffusion analysis indicated a significantly higher crystal existing ratio in the medulla and papilla of SFs and a significantly higher number of papillary crystal deposits in SFs than NSFs. The expression analysis indicated relatively high osteopontin and CD68, low superoxide dismutase, and significantly lower Tamm-Horsfall protein expression levels in SFs. Multivariate logistic regression analysis involving the above factors found the presence of renal papillary crystals as a significant independent factor related to SFs (odds ratio 5.55, 95% confidence interval 1.08-37.18, P = 0.0395).
Conclusions: Regardless of stone formation, intratubular crystals in the renal parenchyma seem to transmigrate to the interstitium. SFs may have reduced ability to eliminate renal parenchymal crystals, particularly those in the papilla region, than NSFs with associated gene expression profiles.
Keywords: Kidney stones; Macrophages; Osteopontin; Oxidative stress; Tamm-Horsfall protein.
Conflict of interest statement
Ethics approval and consent to participate
The present study was carried out with the approval of our intuitional ethics committee (Nagoya City University Hospital, Approved No. 551). All patients’ written consent was obtained prior to the study.
Consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Figures

Similar articles
-
Oxalate absorption and endogenous oxalate synthesis from ascorbate in calcium oxalate stone formers and non-stone formers.Am J Kidney Dis. 2004 Dec;44(6):1060-9. doi: 10.1053/j.ajkd.2004.08.028. Am J Kidney Dis. 2004. PMID: 15558527
-
Genome-Wide Gene Expression Profiling of Randall's Plaques in Calcium Oxalate Stone Formers.J Am Soc Nephrol. 2017 Jan;28(1):333-347. doi: 10.1681/ASN.2015111271. Epub 2016 Jun 13. J Am Soc Nephrol. 2017. PMID: 27297950 Free PMC article.
-
Renal intratubular crystals and hyaluronan staining occur in stone formers with bypass surgery but not with idiopathic calcium oxalate stones.Anat Rec (Hoboken). 2008 Mar;291(3):325-34. doi: 10.1002/ar.20656. Anat Rec (Hoboken). 2008. PMID: 18286613
-
Role of interstitial apatite plaque in the pathogenesis of the common calcium oxalate stone.Semin Nephrol. 2008 Mar;28(2):111-9. doi: 10.1016/j.semnephrol.2008.01.004. Semin Nephrol. 2008. PMID: 18359392 Free PMC article. Review.
-
Insights on the pathology of kidney stone formation.Urol Res. 2005 Nov;33(5):383-9. doi: 10.1007/s00240-005-0488-0. Epub 2005 Aug 3. Urol Res. 2005. PMID: 16078085 Review.
Cited by
-
Effects of secretome derived from macrophages exposed to calcium oxalate crystals on renal fibroblast activation.Commun Biol. 2021 Aug 11;4(1):959. doi: 10.1038/s42003-021-02479-2. Commun Biol. 2021. PMID: 34381146 Free PMC article.
-
Sulfated Laminarin Polysaccharides Reduce the Adhesion of Nano-COM Crystals to Renal Epithelial Cells by Inhibiting Oxidative and Endoplasmic Reticulum Stress.Pharmaceuticals (Basel). 2024 Jun 19;17(6):805. doi: 10.3390/ph17060805. Pharmaceuticals (Basel). 2024. PMID: 38931471 Free PMC article.
-
Renal papillary tip biopsy in stone formers: a review of clinical safety and insights.Urolithiasis. 2024 Jun 18;52(1):93. doi: 10.1007/s00240-024-01596-x. Urolithiasis. 2024. PMID: 38888601 Review.
-
Prevalence and risk factors of kidney stone disease in population aged 40-70 years old in Kharameh cohort study: a cross-sectional population-based study in southern Iran.BMC Urol. 2022 Dec 19;22(1):205. doi: 10.1186/s12894-022-01161-x. BMC Urol. 2022. PMID: 36536352 Free PMC article.
-
Bisphosphonate Use May Reduce the Risk of Urolithiasis in Astronauts on Long-Term Spaceflights.JBMR Plus. 2021 Sep 22;6(1):e10550. doi: 10.1002/jbm4.10550. eCollection 2022 Jan. JBMR Plus. 2021. PMID: 35079672 Free PMC article.
References
Publication types
MeSH terms
Substances
Grants and funding
- 15H04976/Grants-in-Aid for Scientific Research from the Ministry of Education, Culture, Sports, Science, and Technology, Japan
- 15K10627/Grants-in-Aid for Scientific Research from the Ministry of Education, Culture, Sports, Science, and Technology, Japan
- 16K11054/Grants-in-Aid for Scientific Research from the Ministry of Education, Culture, Sports, Science, and Technology, Japan
- 16K15692/Grants-in-Aid for Scientific Research from the Ministry of Education, Culture, Sports, Science, and Technology, Japan
- 16K20153/Grants-in-Aid for Scientific Research from the Ministry of Education, Culture, Sports, Science, and Technology, Japan
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

