Effects and mechanisms of apolipoprotein A-V on the regulation of lipid accumulation in cardiomyocytes
- PMID: 29530023
- PMCID: PMC5848552
- DOI: 10.1186/s12944-018-0692-x
Effects and mechanisms of apolipoprotein A-V on the regulation of lipid accumulation in cardiomyocytes
Abstract
Background: Apolipoprotein (apo) A-V is a key regulator of triglyceride (TG) metabolism. We investigated effects of apoA-V on lipid metabolism in cardiomyocytes in this study.
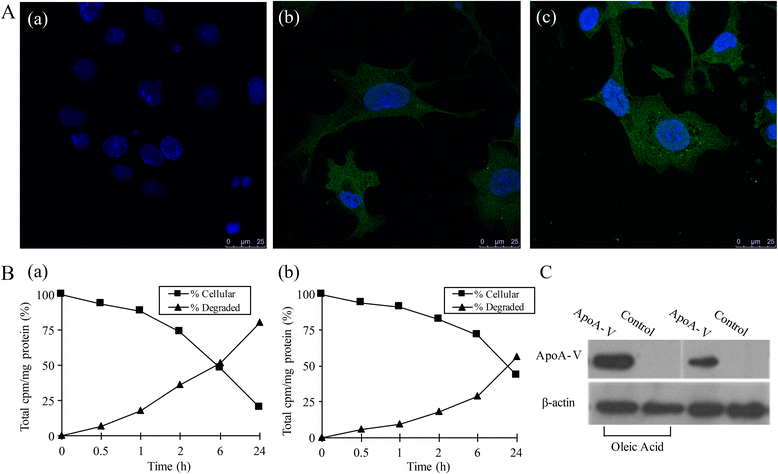
Methods: We first examined whether apoA-V can be taken up by cardiomyocytes and whether low density lipoprotein receptor family members participate in this process. Next, triglyceride (TG) content and lipid droplet changes were detected at different concentrations of apoA-V in normal and lipid-accumulation cells in normal and obese animals. Finally, we tested the levels of fatty acids (FAs) taken up into cardiomyocytes and lipid secretion through [14C]-oleic acid.
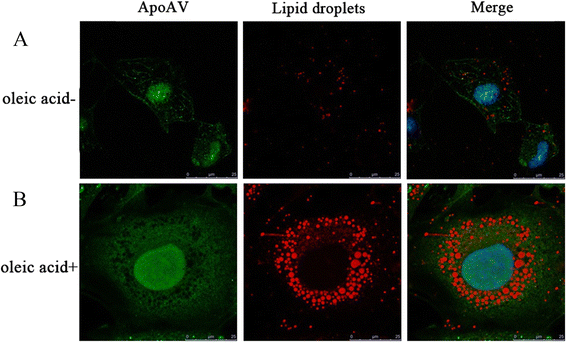
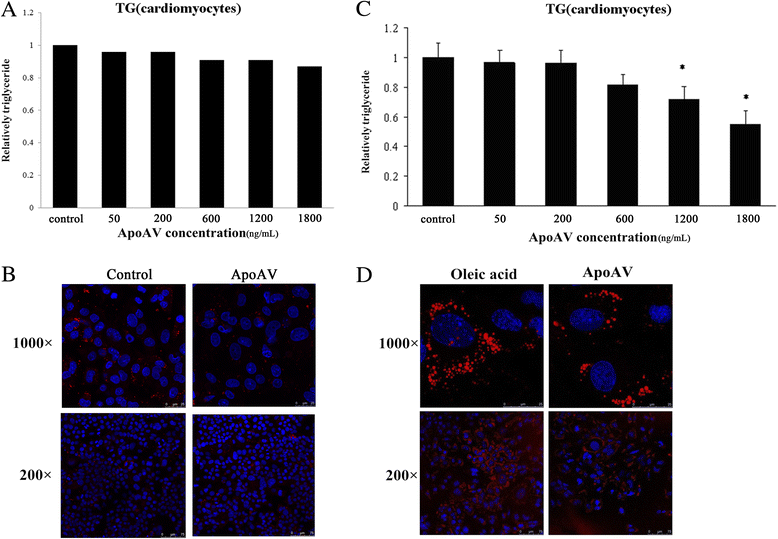
Results: Our results show that heart tissue has apoA-V protein, and apoA-V is taken up by cardiomyocytes. When HL-1 cells were transfected with low density lipoprotein receptor (LDLR)-related protein 1(LRP1) siRNA, apoA-V intake decreased by 53% (P<0.05), while a 37% lipid accumulation in HL-1 cells remain unchanged. ApoA-V localized to the cytoplasm and was associated with lipid droplets in HL-1 cells. A 1200 and 1800 ng/mL apoA-V intervention decreased TG content by 28% and 45% in HL-1 cells, respectively and decreased TG content by 39% in mouse heart tissue (P<0.05). However, apoA-V had no effects on TG content in either normal HL-1 cells or mice. The levels of FAs taken up into cardiomyocytes decreased by 43% (P < 0.05), and the levels of TG and cholesterol ester secretion increased by 1.2-fold and 1.6-fold, respectively (P < 0.05).
Conclusion: ApoA-V is a novel regulator of lipid metabolism in cardiomyocytes.
Keywords: Apolipoprotein A-V; HL-1 cells; Lipid droplets; Triglyceride.
Conflict of interest statement
Ethics approval and consent to participate
Not applicable
Consent for publication
Not applicable
Competing interests
The authors declare that they have no competing interests.
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Figures






Similar articles
-
Apolipoprotein A-IV expression in mouse liver enhances triglyceride secretion and reduces hepatic lipid content by promoting very low density lipoprotein particle expansion.Arterioscler Thromb Vasc Biol. 2013 Nov;33(11):2501-8. doi: 10.1161/ATVBAHA.113.301948. Epub 2013 Sep 12. Arterioscler Thromb Vasc Biol. 2013. PMID: 24030551
-
Apolipoprotein A-V associates with intrahepatic lipid droplets and influences triglyceride accumulation.Biochim Biophys Acta. 2010 May;1801(5):605-8. doi: 10.1016/j.bbalip.2010.02.004. Epub 2010 Feb 11. Biochim Biophys Acta. 2010. PMID: 20153840 Free PMC article.
-
Low-density lipoprotein receptor-related protein 1 mediates hypoxia-induced very low density lipoprotein-cholesteryl ester uptake and accumulation in cardiomyocytes.Cardiovasc Res. 2012 Jun 1;94(3):469-79. doi: 10.1093/cvr/cvs136. Epub 2012 Mar 27. Cardiovasc Res. 2012. PMID: 22454363
-
Apolipoprotein A-V dependent modulation of plasma triacylglycerol: a puzzlement.Biochim Biophys Acta. 2012 May;1821(5):795-9. doi: 10.1016/j.bbalip.2011.12.002. Epub 2011 Dec 22. Biochim Biophys Acta. 2012. PMID: 22209939 Free PMC article. Review.
-
Influence of apolipoprotein A-V on the metabolic fate of triacylglycerol.Curr Opin Lipidol. 2013 Apr;24(2):153-9. doi: 10.1097/MOL.0b013e32835c8c1a. Curr Opin Lipidol. 2013. PMID: 23241513 Free PMC article. Review.
Cited by
-
Apolipoprotein A5 ameliorates MCT induced pulmonary hypertension by inhibiting ER stress in a GRP78 dependent mechanism.Lipids Health Dis. 2022 Aug 8;21(1):69. doi: 10.1186/s12944-022-01680-4. Lipids Health Dis. 2022. PMID: 35941581 Free PMC article.
-
Dissecting the Gene Expression Networks Associated with Variations in the Major Components of the Fatty Acid Semimembranosus Muscle Profile in Large White Heavy Pigs.Animals (Basel). 2021 Feb 27;11(3):628. doi: 10.3390/ani11030628. Animals (Basel). 2021. PMID: 33673460 Free PMC article.
References
-
- van der Vliet HN, Schaap FG, Levels JH, Ottenhoff R, Looije N, Wesseling JG, Groen AK, Chamuleau RA. Adenoviral overexpression of apolipoprotein A-V reduces serum levels of triglycerides and cholesterol in mice. Biochem Biophys Res Commun. 2002;295:1156–1159. doi: 10.1016/S0006-291X(02)00808-2. - DOI - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

