Seasonal Reproduction in Vertebrates: Melatonin Synthesis, Binding, and Functionality Using Tinbergen's Four Questions
- PMID: 29534047
- PMCID: PMC6017951
- DOI: 10.3390/molecules23030652
Seasonal Reproduction in Vertebrates: Melatonin Synthesis, Binding, and Functionality Using Tinbergen's Four Questions
Abstract
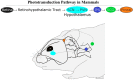
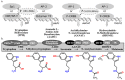
One of the many functions of melatonin in vertebrates is seasonal reproductive timing. Longer nights in winter correspond to an extended duration of melatonin secretion. The purpose of this review is to discuss melatonin synthesis, receptor subtypes, and function in the context of seasonality across vertebrates. We conclude with Tinbergen's Four Questions to create a comparative framework for future melatonin research in the context of seasonal reproduction.
Keywords: melatonin; seasonal reproduction; vertebrates.
Conflict of interest statement
The Authors declare that there is no conflict of interest.
Figures




References
-
- Tan D.-X., Manchester L.C., Liu X., Rosales-Corral S.A., Acuna-Castroviejo D., Reiter R.J. Mitochondria and chloroplasts as the original sites of melatonin synthesis: A hypothesis related to melatonin’s primary function and evolution in eukaryotes. J. Pineal Res. 2013;54:127–138. doi: 10.1111/jpi.12026. - DOI - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

