Mycophenolic acid induces senescence of vascular precursor cells
- PMID: 29538431
- PMCID: PMC5851606
- DOI: 10.1371/journal.pone.0193749
Mycophenolic acid induces senescence of vascular precursor cells
Abstract
Objective: Endothelial dysfunction is central to the pathogenesis of many rheumatic diseases, typified by vascular inflammation and damage. Immunosuppressive drugs induce disease remission and lead to improved patient survival. However, there remains a higher incidence of cardiovascular disease in these patients even after adequate disease control. The purpose of this study was to determine the effect of mycophenolic acid (MPA), a commonly used immunosuppressive drug in rheumatology, on blood vessel or circulating endothelial colony forming cell number and function.
Methods: We tested whether mycophenolic acid exerts an inhibitory effect on proliferation, clonogenic potential and vasculogenic function of endothelial colony forming cell. We also studied potential mechanisms involved in the observed effects.
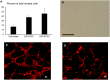
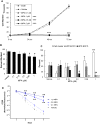
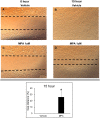
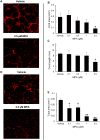
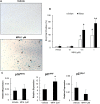
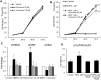
Results: Treatment with MPA decreased endothelial colony forming cell proliferation, clonogenic potential and vasculogenic function in a dose-dependent fashion. MPA increased senescence-associated β-galactosidase expression, p21 gene expression and p53 phosphorylation, indicative of activation of cellular senescence. Exogenous guanosine supplementation rescued diminished endothelial colony forming cell proliferation and indices of senescence, consistent with the known mechanism of action of MPA.
Conclusion: Our findings show that clinically relevant doses of MPA have potent anti-angiogenic and pro-senescent effects on vascular precursor cells in vitro, thus indicating that treatment with MPA can potentially affect vascular repair and regeneration. This warrants further studies in vivo to determine how MPA therapy contributes to vascular dysfunction and increased cardiovascular disease seen in patients with inflammatory rheumatic disease.
Conflict of interest statement
Figures
Similar articles
-
Interferon-gamma induces cellular senescence through p53-dependent DNA damage signaling in human endothelial cells.Mech Ageing Dev. 2009 Mar;130(3):179-88. doi: 10.1016/j.mad.2008.11.004. Epub 2008 Nov 21. Mech Ageing Dev. 2009. PMID: 19071156
-
Suppression of Transforming Growth Factor-β Signaling Delays Cellular Senescence and Preserves the Function of Endothelial Cells Derived from Human Pluripotent Stem Cells.Stem Cells Transl Med. 2017 Feb;6(2):589-600. doi: 10.5966/sctm.2016-0089. Epub 2016 Sep 20. Stem Cells Transl Med. 2017. PMID: 28191769 Free PMC article.
-
Activation of PPARγ/P53 signaling is required for curcumin to induce hepatic stellate cell senescence.Cell Death Dis. 2016 Apr 14;7(4):e2189. doi: 10.1038/cddis.2016.92. Cell Death Dis. 2016. PMID: 27077805 Free PMC article.
-
Mycophenolic acid in rheumatology: mechanisms of action and severe adverse events.Reumatismo. 2010 Apr-Jun;62(2):91-100. doi: 10.4081/reumatismo.2010.91. Reumatismo. 2010. PMID: 20657885 Review.
-
Senescence and its bypass in the vascular endothelium.Front Biosci. 2005 Jan 1;10:940-50. doi: 10.2741/1588. Print 2005 Jan 1. Front Biosci. 2005. PMID: 15569632 Review.
Cited by
-
Premature senescence of placental decidua cells as a possible cause of miscarriage produced by mycophenolic acid.J Biomed Sci. 2021 Jan 4;28(1):3. doi: 10.1186/s12929-020-00704-4. J Biomed Sci. 2021. PMID: 33397374 Free PMC article.
-
Titer improvement of mycophenolic acid in the novel producer strain Penicillium arizonense and expression analysis of its biosynthetic genes.BMC Microbiol. 2023 May 17;23(1):135. doi: 10.1186/s12866-023-02884-z. BMC Microbiol. 2023. PMID: 37198535 Free PMC article.
-
Combining multi-omics analysis to identify host-targeted targets for the control of Brucella infection.Microb Biotechnol. 2023 Dec;16(12):2345-2366. doi: 10.1111/1751-7915.14307. Epub 2023 Oct 26. Microb Biotechnol. 2023. PMID: 37882474 Free PMC article.
References
-
- Gelber A, Levine S, Rosen A. Gelber A.C., Levine S.M., Gelber Rosen A, Allan C., et al.Inflammatory Rheumatic Diseases In: Hammer G, McPhee S. Hammer G.D., McPhee S.J. Eds. Hammer Gary D., and McPhee Stephen J.. eds. Pathophysiology of Disease: An Introduction to Clinical Medicine, Seventh Edition New York, NY: McGraw-Hill; 2013.
-
- Urowitz MB, Bookman AA, Koehler BE, Gordon DA, Smythe HA, Ogryzlo MA. The bimodal mortality pattern of systemic lupus erythematosus. Am J Med 1976;60:221–225. - PubMed
-
- Nossent J, Cikes N, Kiss E, Marchesoni A, Nassanova V, Mosca M, et al. Current causes of death in systemic lupus erythematosus in Europe, 2000–2004: relation to disease activity and damage accrual. Lupus 2007;16:309–317 doi: 10.1177/0961203307077987 - DOI - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

