Immune-related adverse events for anti-PD-1 and anti-PD-L1 drugs: systematic review and meta-analysis
- PMID: 29540345
- PMCID: PMC5851471
- DOI: 10.1136/bmj.k793
Immune-related adverse events for anti-PD-1 and anti-PD-L1 drugs: systematic review and meta-analysis
Abstract
Objective: To evaluate rates of serious organ specific immune-related adverse events, general adverse events related to immune activation, and adverse events consistent with musculoskeletal problems for anti-programmed cell death 1 (PD-1) drugs overall and compared with control treatments.
Design: Systematic review and meta-analysis.
Data sources: Medline, Embase, Cochrane Library, Web of Science, and Scopus searched to 16 March 2017 and combined with data from ClinicalTrials.gov.
Study selection: Eligible studies included primary clinical trial data on patients with cancer with recurrent or metastatic disease.
Data extraction: Three independent investigators extracted data on adverse events from ClinicalTrials.gov and the published studies. Risk of bias was assessed using the Cochrane tool by three independent investigators.
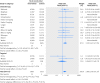
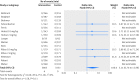
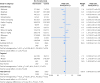
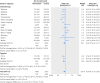
Results: 13 relevant studies were included; adverse event data were available on ClinicalTrials.gov for eight. Studies compared nivolumab (n=6), pembrolizumab (5), or atezolizumab (2) with chemotherapy (11), targeted drugs (1), or both (1). Serious organ specific immune-related adverse events were rare, but compared with standard treatment, rates of hypothyroidism (odds ratio 7.56, 95% confidence interval 4.53 to 12.61), pneumonitis (5.37, 2.73 to 10.56), colitis (2.88, 1.30 to 6.37), and hypophysitis (3.38, 1.02 to 11.08) were increased with anti-PD-1 drugs. Of the general adverse events related to immune activation, only the rate of rash (2.34, 2.73 to 10.56) increased. Incidence of fatigue (32%) and diarrhea (19%) were high but similar to control. Reporting of adverse events consistent with musculoskeletal problems was inconsistent; rates varied but were over 20% in some studies for arthraligia and back pain.
Conclusions: Organ specific immune-related adverse events are uncommon with anti-PD-1 drugs but the risk is increased compared with control treatments. General adverse events related to immune activation are largely similar. Adverse events consistent with musculoskeletal problems are inconsistently reported but adverse events may be common.
Published by the BMJ Publishing Group Limited. For permission to use (where not already granted under a licence) please go to http://group.bmj.com/group/rights-licensing/permissions.
Conflict of interest statement
Competing interests: All authors have completed the ICMJE uniform disclosure form at www.icmje.org/coi_disclosure.pdf and declare: no support from any organization for the submitted work; no financial relationships with any organisations that might have an interest in the submitted work in the previous three years; no other relationships or activities that could appear to have influenced the submitted work.
Figures





References
-
- IMS Institute for Healthcare Informatics. Global Use of Medicines: Outlook through 2017. 2017. http://www.quotidianosanita.it/allegati/allegato1501906.pdf
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
