PcxL and HpxL are flavin-dependent, oxime-forming N-oxidases in phosphonocystoximic acid biosynthesis in Streptomyces
- PMID: 29540479
- PMCID: PMC5936822
- DOI: 10.1074/jbc.RA118.001721
PcxL and HpxL are flavin-dependent, oxime-forming N-oxidases in phosphonocystoximic acid biosynthesis in Streptomyces
Abstract
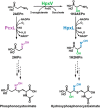
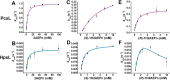
Several oxime-containing small molecules have useful properties, including antimicrobial, insecticidal, anticancer, and immunosuppressive activities. Phosphonocystoximate and its hydroxylated congener, hydroxyphosphonocystoximate, are recently discovered oxime-containing natural products produced by Streptomyces sp. NRRL S-481 and Streptomyces regensis NRRL WC-3744, respectively. The biosynthetic pathways for these two compounds are proposed to diverge at an early step in which 2-aminoethylphosphonate (2AEPn) is converted to (S)-1-hydroxy-2-aminoethylphosphonate ((S)-1H2AEPn) in S. regensis but not in Streptomyces sp. NRRL S-481). Subsequent installation of the oxime moiety into either 2AEPn or (S)-1H2AEPn is predicted to be catalyzed by PcxL or HpxL from Streptomyces sp. NRRL S-481 and S. regensis NRRL WC-3744, respectively, whose sequence and predicted structural characteristics suggest they are unusual N-oxidases. Here, we show that recombinant PcxL and HpxL catalyze the FAD- and NADPH-dependent oxidation of 2AEPn and 1H2AEPn, producing a mixture of the respective aldoximes and nitrosylated phosphonic acid products. Measurements of catalytic efficiency indicated that PcxL has almost an equal preference for 2AEPn and (R)-1H2AEPn. 2AEPn was turned over at a 10-fold higher rate than (R)-1H2AEPn under saturating conditions, resulting in a similar but slightly lower kcat/Km We observed that (S)-1H2AEPn is a relatively poor substrate for PcxL but is clearly the preferred substrate for HpxL, consistent with the proposed biosynthetic pathway in S. regensis. HpxL also used both 2AEPn and (R)-1H2AEPn, with the latter inhibiting HpxL at high concentrations. Bioinformatic analysis indicated that PcxL and HpxL are members of a new class of oxime-forming N-oxidases that are broadly dispersed among bacteria.
Keywords: enzyme kinetics; flavin adenine dinucleotide (FAD); natural product biosynthesis; nicotinamide; oxidase; oxime; phosphonates; phosphonic acid; substrate specificity.
© 2018 Goettge et al.
Conflict of interest statement
The authors declare that they have no conflicts of interest with the contents of this article
Figures



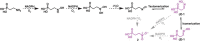
Similar articles
-
Characterization of a broadly specific cadaverine N-hydroxylase involved in desferrioxamine B biosynthesis in Streptomyces sviceus.PLoS One. 2021 Mar 30;16(3):e0248385. doi: 10.1371/journal.pone.0248385. eCollection 2021. PLoS One. 2021. PMID: 33784308 Free PMC article.
-
Structural analyses of the Group A flavin-dependent monooxygenase PieE reveal a sliding FAD cofactor conformation bridging OUT and IN conformations.J Biol Chem. 2020 Apr 3;295(14):4709-4722. doi: 10.1074/jbc.RA119.011212. Epub 2020 Feb 28. J Biol Chem. 2020. PMID: 32111738 Free PMC article.
-
Characterization of SgcE6, the flavin reductase component supporting FAD-dependent halogenation and hydroxylation in the biosynthesis of the enediyne antitumor antibiotic C-1027.FEMS Microbiol Lett. 2009 Nov;300(2):237-41. doi: 10.1111/j.1574-6968.2009.01802.x. Epub 2009 Sep 27. FEMS Microbiol Lett. 2009. PMID: 19817865 Free PMC article.
-
Mechanisms of reduced flavin transfer in the two-component flavin-dependent monooxygenases.Arch Biochem Biophys. 2014 Aug;555-556:33-46. doi: 10.1016/j.abb.2014.05.009. Epub 2014 May 20. Arch Biochem Biophys. 2014. PMID: 24857824 Review.
-
Biosynthesis of phosphonic and phosphinic acid natural products.Annu Rev Biochem. 2009;78:65-94. doi: 10.1146/annurev.biochem.78.091707.100215. Annu Rev Biochem. 2009. PMID: 19489722 Free PMC article. Review.
Cited by
-
A biosynthetic aspartate N-hydroxylase performs successive oxidations by holding intermediates at a site away from the catalytic center.J Biol Chem. 2023 Jul;299(7):104904. doi: 10.1016/j.jbc.2023.104904. Epub 2023 Jun 10. J Biol Chem. 2023. PMID: 37302552 Free PMC article.
-
A Novel Biosynthetic Gene Cluster Across the Pantoea Species Complex Is Important for Pathogenicity in Onion.Mol Plant Microbe Interact. 2023 Mar;36(3):176-188. doi: 10.1094/MPMI-08-22-0165-R. Epub 2023 Mar 9. Mol Plant Microbe Interact. 2023. PMID: 36534063 Free PMC article.
-
Genomic context analysis enables the discovery of an unusual NAD-dependent racemase in phosphonate catabolism.FEBS J. 2025 Aug;292(16):4272-4288. doi: 10.1111/febs.70130. Epub 2025 May 19. FEBS J. 2025. PMID: 40384479 Free PMC article.
References
-
- Bacher M., Brader G., Hofer O., and Greger H. (1999) Oximes from seeds of Atalantia ceylanica. Phytochemistry 50, 991–994 10.1016/S0031-9422(98)00638-4 - DOI
-
- Moya P., Castillo M., Primo-Yúfera E., Couillaud F., Martínez-Máñez R., Garcerá M. D., Miranda M. A., Primo J., and Martínez-Pardo R. (1997) Brevioxime: A new juvenile hormone biosynthesis inhibitor isolated from Penicillium brevicompactum. J. Org. Chem. 62, 8544–8545 10.1021/jo970397y - DOI - PubMed
Publication types
MeSH terms
Substances
Associated data
- Actions
- Actions
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

