Distinct prophase arrest mechanisms in human male meiosis
- PMID: 29540502
- PMCID: PMC6124541
- DOI: 10.1242/dev.160614
Distinct prophase arrest mechanisms in human male meiosis
Abstract
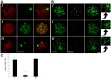
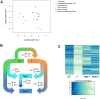
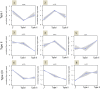
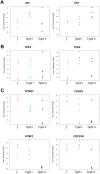
To prevent chromosomal aberrations being transmitted to the offspring, strict meiotic checkpoints are in place to remove aberrant spermatocytes. However, in about 1% of males these checkpoints cause complete meiotic arrest leading to azoospermia and subsequent infertility. Here, we unravel two clearly distinct meiotic arrest mechanisms that occur during prophase of human male meiosis. Type I arrested spermatocytes display severe asynapsis of the homologous chromosomes, disturbed XY-body formation and increased expression of the Y chromosome-encoded gene ZFY and seem to activate a DNA damage pathway leading to induction of p63, possibly causing spermatocyte apoptosis. Type II arrested spermatocytes display normal chromosome synapsis, normal XY-body morphology and meiotic crossover formation but have a lowered expression of several cell cycle regulating genes and fail to silence the X chromosome-encoded gene ZFX Discovery and understanding of these meiotic arrest mechanisms increases our knowledge of how genomic stability is guarded during human germ cell development.
Keywords: Human spermatogenesis; Infertility; Meiosis; Meiotic arrest; Meiotic silencing; p63.
© 2018. Published by The Company of Biologists Ltd.
Conflict of interest statement
Competing interestsThe authors declare no competing or financial interests.
Figures


Similar articles
-
Meiotic defects and decreased expression of genes located around the chromosomal breakpoint in the testis of a patient with a novel 46,X,t(Y;1)(p11.3;p31) translocation.Int J Mol Med. 2017 Aug;40(2):367-377. doi: 10.3892/ijmm.2017.3029. Epub 2017 Jun 14. Int J Mol Med. 2017. PMID: 28627638 Free PMC article.
-
Meiotic Chromosome Synapsis and XY-Body Formation In Vitro.Front Endocrinol (Lausanne). 2021 Oct 14;12:761249. doi: 10.3389/fendo.2021.761249. eCollection 2021. Front Endocrinol (Lausanne). 2021. PMID: 34721307 Free PMC article.
-
ATM and RPA in meiotic chromosome synapsis and recombination.Nat Genet. 1997 Dec;17(4):457-61. doi: 10.1038/ng1297-457. Nat Genet. 1997. PMID: 9398850
-
MEIOSIN directs initiation of meiosis and subsequent meiotic prophase program during spermatogenesis.Genes Genet Syst. 2022 Jun 4;97(1):27-39. doi: 10.1266/ggs.21-00054. Epub 2021 Dec 25. Genes Genet Syst. 2022. PMID: 34955498 Review.
-
In vitro spermatogenesis: Why meiotic checkpoints matter.Curr Top Dev Biol. 2023;151:345-369. doi: 10.1016/bs.ctdb.2022.04.009. Epub 2022 Jun 9. Curr Top Dev Biol. 2023. PMID: 36681476 Review.
Cited by
-
Transcriptional progression during meiotic prophase I reveals sex-specific features and X chromosome dynamics in human fetal female germline.PLoS Genet. 2021 Sep 9;17(9):e1009773. doi: 10.1371/journal.pgen.1009773. eCollection 2021 Sep. PLoS Genet. 2021. PMID: 34499650 Free PMC article.
-
In Vitro Meiosis of Male Germline Stem Cells.Stem Cell Reports. 2020 Nov 10;15(5):1140-1153. doi: 10.1016/j.stemcr.2020.10.006. Stem Cell Reports. 2020. PMID: 33176123 Free PMC article.
-
Disruptive natural selection by male reproductive potential prevents underexpression of protein-coding genes on the human Y chromosome as a self-domestication syndrome.BMC Genet. 2020 Oct 22;21(Suppl 1):89. doi: 10.1186/s12863-020-00896-6. BMC Genet. 2020. PMID: 33092533 Free PMC article.
-
Understanding the Underlying Molecular Mechanisms of Meiotic Arrest during In Vitro Spermatogenesis in Rat Prepubertal Testicular Tissue.Int J Mol Sci. 2022 May 24;23(11):5893. doi: 10.3390/ijms23115893. Int J Mol Sci. 2022. PMID: 35682573 Free PMC article.
-
Mate-pair genome sequencing reveals structural variants for idiopathic male infertility.Hum Genet. 2023 Mar;142(3):363-377. doi: 10.1007/s00439-022-02510-4. Epub 2022 Dec 16. Hum Genet. 2023. PMID: 36526900
References
-
- Barchi M., Mahadevaiah S., Di Giacomo M., Baudat F., De Rooij D. G., Burgoyne P. S., Jasin M. and Keeney S. (2005). Surveillance of different recombination defects in mouse spermatocytes yields distinct responses despite elimination at an identical developmental stage. Mol. Cell. Biol. 25, 7203-7215. 10.1128/MCB.25.16.7203-7215.2005 - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

