Obesity promotes resistance to anti-VEGF therapy in breast cancer by up-regulating IL-6 and potentially FGF-2
- PMID: 29540614
- PMCID: PMC5936748
- DOI: 10.1126/scitranslmed.aag0945
Obesity promotes resistance to anti-VEGF therapy in breast cancer by up-regulating IL-6 and potentially FGF-2
Abstract
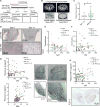
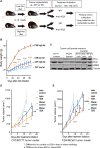
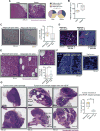
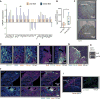
Anti-vascular endothelial growth factor (VEGF) therapy has failed to improve survival in patients with breast cancer (BC). Potential mechanisms of resistance to anti-VEGF therapy include the up-regulation of alternative angiogenic and proinflammatory factors. Obesity is associated with hypoxic adipose tissues, including those in the breast, resulting in increased production of some of the aforementioned factors. Hence, we hypothesized that obesity could contribute to anti-VEGF therapy's lack of efficacy. We found that BC patients with obesity harbored increased systemic concentrations of interleukin-6 (IL-6) and/or fibroblast growth factor 2 (FGF-2), and their tumor vasculature was less sensitive to anti-VEGF treatment. Mouse models revealed that obesity impairs the effects of anti-VEGF on angiogenesis, tumor growth, and metastasis. In one murine BC model, obesity was associated with increased IL-6 production from adipocytes and myeloid cells within tumors. IL-6 blockade abrogated the obesity-induced resistance to anti-VEGF therapy in primary and metastatic sites by directly affecting tumor cell proliferation, normalizing tumor vasculature, alleviating hypoxia, and reducing immunosuppression. Similarly, in a second mouse model, where obesity was associated with increased FGF-2, normalization of FGF-2 expression by metformin or specific FGF receptor inhibition decreased vessel density and restored tumor sensitivity to anti-VEGF therapy in obese mice. Collectively, our data indicate that obesity fuels BC resistance to anti-VEGF therapy via the production of inflammatory and angiogenic factors.
Copyright © 2018 The Authors, some rights reserved; exclusive licensee American Association for the Advancement of Science. No claim to original U.S. Government Works.
Conflict of interest statement
Figures


Comment in
-
Breast cancer: Obesity - tipping the scales of resistance.Nat Rev Cancer. 2018 May;18(5):265. doi: 10.1038/nrc.2018.31. Epub 2018 Apr 6. Nat Rev Cancer. 2018. PMID: 29610490 No abstract available.
-
Obesity associated with resistance to anti-angiogenic therapy.Nat Rev Clin Oncol. 2018 Jun;15(6):344. doi: 10.1038/s41571-018-0011-5. Nat Rev Clin Oncol. 2018. PMID: 29626201 No abstract available.
-
Anti-VEGF therapy - a role in obesity-related breast cancer.Nat Rev Endocrinol. 2018 Jun;14(6):329-330. doi: 10.1038/s41574-018-0021-5. Nat Rev Endocrinol. 2018. PMID: 29695750 Free PMC article.
-
Obesity Protects Cancer from Drugs Targeting Blood Vessels.Cell Metab. 2018 Jun 5;27(6):1163-1165. doi: 10.1016/j.cmet.2018.05.014. Cell Metab. 2018. PMID: 29874563
Similar articles
-
Dual blockade of vascular endothelial growth factor (VEGF) and basic fibroblast growth factor (FGF-2) exhibits potent anti-angiogenic effects.Cancer Lett. 2016 Jul 28;377(2):164-73. doi: 10.1016/j.canlet.2016.04.036. Epub 2016 Apr 26. Cancer Lett. 2016. PMID: 27130666
-
Distinct role of fibroblast growth factor-2 and vascular endothelial growth factor on tumor growth and angiogenesis.Am J Pathol. 2003 Jun;162(6):1913-26. doi: 10.1016/S0002-9440(10)64325-8. Am J Pathol. 2003. PMID: 12759248 Free PMC article.
-
Therapeutic paradigm of dual targeting VEGF and PDGF for effectively treating FGF-2 off-target tumors.Nat Commun. 2020 Jul 24;11(1):3704. doi: 10.1038/s41467-020-17525-6. Nat Commun. 2020. PMID: 32709869 Free PMC article.
-
Anti-FGF2 approaches as a strategy to compensate resistance to anti-VEGF therapy: long-pentraxin 3 as a novel antiangiogenic FGF2-antagonist.Eur Cytokine Netw. 2009 Dec;20(4):225-34. doi: 10.1684/ecn.2009.0175. Eur Cytokine Netw. 2009. PMID: 20167562 Review.
-
Mechanisms of resistance to vascular endothelial growth factor blockade.Cancer. 2012 Jul 15;118(14):3455-67. doi: 10.1002/cncr.26540. Epub 2011 Nov 15. Cancer. 2012. PMID: 22086782 Review.
Cited by
-
Obesity and leptin in breast cancer angiogenesis.Front Endocrinol (Lausanne). 2024 Oct 8;15:1465727. doi: 10.3389/fendo.2024.1465727. eCollection 2024. Front Endocrinol (Lausanne). 2024. PMID: 39439572 Free PMC article. Review.
-
Interleukin-6-mediated epigenetic control of the VEGFR2 gene induces disorganized angiogenesis in human breast tumors.J Biol Chem. 2020 Aug 21;295(34):12086-12098. doi: 10.1074/jbc.RA120.012590. Epub 2020 Jul 7. J Biol Chem. 2020. PMID: 32636303 Free PMC article.
-
Elevated extracellular matrix protein 1 in circulating extracellular vesicles supports breast cancer progression under obesity conditions.Nat Commun. 2024 Feb 24;15(1):1685. doi: 10.1038/s41467-024-45995-5. Nat Commun. 2024. PMID: 38402239 Free PMC article.
-
Escape from breast tumor dormancy: The convergence of obesity and menopause.Proc Natl Acad Sci U S A. 2022 Oct 11;119(41):e2204758119. doi: 10.1073/pnas.2204758119. Epub 2022 Oct 3. Proc Natl Acad Sci U S A. 2022. PMID: 36191215 Free PMC article.
-
Dissecting the emerging role of cancer-associated adipocyte-derived cytokines in remodeling breast cancer progression.Heliyon. 2024 Jul 26;10(15):e35200. doi: 10.1016/j.heliyon.2024.e35200. eCollection 2024 Aug 15. Heliyon. 2024. PMID: 39161825 Free PMC article. Review.
References
-
- Lohmann AE, Chia S. Patients with metastatic breast cancer using bevacizumab as a treatment: Is there still a role for it? Curr Treat Options Oncol. 2012;13:249–262. - PubMed
-
- Guiu B, Petit JM, Bonnetain F, Ladoire S, Guiu S, Cercueil JP, Krausé D, Hillon P, Borg C, Chauffert B, Ghiringhelli F. Visceral fat area is an independent predictive biomarker of outcome after first-line bevacizumab-based treatment in metastatic colorectal cancer. Gut. 2010;59:341–347. - PubMed
-
- Ladoire S, Bonnetain F, Gauthier M, Zanetta S, Petit JM, Guiu S, Kermarrec I, Mourey E, Michel F, Krause D, Hillon P, Cormier L, Ghiringhelli F, Guiu B. Visceral fat area as a new independent predictive factor of survival in patients with metastatic renal cell carcinoma treated with antiangiogenic agents. Oncologist. 2011;16:71–81. - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

