Use of a Tyrosine Analogue To Modulate the Two Activities of a Nonheme Iron Enzyme OvoA in Ovothiol Biosynthesis, Cysteine Oxidation versus Oxidative C-S Bond Formation
- PMID: 29544051
- PMCID: PMC5884719
- DOI: 10.1021/jacs.7b13628
Use of a Tyrosine Analogue To Modulate the Two Activities of a Nonheme Iron Enzyme OvoA in Ovothiol Biosynthesis, Cysteine Oxidation versus Oxidative C-S Bond Formation
Abstract
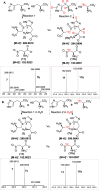
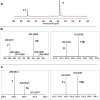
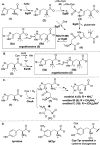
Ovothiol is a histidine thiol derivative. The biosynthesis of ovothiol involves an extremely efficient trans-sulfuration strategy. The nonheme iron enzyme OvoA catalyzed oxidative coupling between cysteine and histidine is one of the key steps. Besides catalyzing the oxidative coupling between cysteine and histidine, OvoA also catalyzes the oxidation of cysteine to cysteine sulfinic acid (cysteine dioxygenase activity). Thus far, very little mechanistic information is available for OvoA-catalysis. In this report, we measured the kinetic isotope effect (KIE) in OvoA-catalysis using the isotopically sensitive branching method. In addition, by replacing an active site tyrosine (Tyr417) with 2-amino-3-(4-hydroxy-3-(methylthio)phenyl)propanoic acid (MtTyr) through the amber suppressor mediated unnatural amino acid incorporation method, the two OvoA activities (oxidative coupling between cysteine and histidine, and cysteine dioxygenase activity) can be modulated. These results suggest that the two OvoA activities branch out from a common intermediate and that the active site tyrosine residue plays some key roles in controlling the partitioning between these two pathways.
Figures
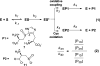
Similar articles
-
Biochemical and Structural Characterization of OvoATh2: A Mononuclear Nonheme Iron Enzyme from Hydrogenimonas thermophila for Ovothiol Biosynthesis.ACS Catal. 2023 Nov 14;13(23):15417-15426. doi: 10.1021/acscatal.3c04026. eCollection 2023 Dec 1. ACS Catal. 2023. PMID: 38058600 Free PMC article.
-
Cysteine oxidation reactions catalyzed by a mononuclear non-heme iron enzyme (OvoA) in ovothiol biosynthesis.Org Lett. 2014 Apr 18;16(8):2122-5. doi: 10.1021/ol5005438. Epub 2014 Mar 31. Org Lett. 2014. PMID: 24684381 Free PMC article.
-
Regioselectivity of the oxidative C-S bond formation in ergothioneine and ovothiol biosyntheses.Org Lett. 2013 Sep 20;15(18):4854-7. doi: 10.1021/ol402275t. Epub 2013 Sep 9. Org Lett. 2013. PMID: 24016264 Free PMC article.
-
New Frontiers in Nonheme Enzymatic Oxyferryl Species.Chembiochem. 2024 Nov 18;25(22):e202400307. doi: 10.1002/cbic.202400307. Epub 2024 Aug 7. Chembiochem. 2024. PMID: 38900645 Free PMC article. Review.
-
Bio-inspired Nonheme Iron Oxidation Catalysis: Involvement of Oxoiron(V) Oxidants in Cleaving Strong C-H Bonds.Angew Chem Int Ed Engl. 2020 May 4;59(19):7332-7349. doi: 10.1002/anie.201906551. Epub 2020 Mar 2. Angew Chem Int Ed Engl. 2020. PMID: 31373120 Review.
Cited by
-
Crystal Structure of the Ergothioneine Sulfoxide Synthase from Candidatus Chloracidobacterium thermophilum and Structure-Guided Engineering To Modulate Its Substrate Selectivity.ACS Catal. 2019 Aug 2;9(8):6955-6961. doi: 10.1021/acscatal.9b02054. Epub 2019 Jul 2. ACS Catal. 2019. PMID: 32257583 Free PMC article.
-
Chemical modifications of proteins and their applications in metalloenzyme studies.Synth Syst Biotechnol. 2021 Feb 15;6(1):32-49. doi: 10.1016/j.synbio.2021.01.001. eCollection 2021 Mar. Synth Syst Biotechnol. 2021. PMID: 33665390 Free PMC article. Review.
-
Noncanonical Amino Acids: Bringing New-to-Nature Functionalities to Biocatalysis.Chem Rev. 2024 Oct 9;124(19):10877-10923. doi: 10.1021/acs.chemrev.4c00136. Epub 2024 Sep 27. Chem Rev. 2024. PMID: 39329413 Free PMC article. Review.
-
Biochemical and Structural Characterization of OvoATh2: A Mononuclear Nonheme Iron Enzyme from Hydrogenimonas thermophila for Ovothiol Biosynthesis.ACS Catal. 2023 Nov 14;13(23):15417-15426. doi: 10.1021/acscatal.3c04026. eCollection 2023 Dec 1. ACS Catal. 2023. PMID: 38058600 Free PMC article.
-
OvoAMtht from Methyloversatilis thermotolerans ovothiol biosynthesis is a bifunction enzyme: thiol oxygenase and sulfoxide synthase activities.Chem Sci. 2022 Mar 2;13(12):3589-3598. doi: 10.1039/d1sc05479a. eCollection 2022 Mar 24. Chem Sci. 2022. PMID: 35432880 Free PMC article.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

