Development and validation of a diagnostic model for early differentiation of sepsis and non-infectious SIRS in critically ill children - a data-driven approach using machine-learning algorithms
- PMID: 29544449
- PMCID: PMC5853156
- DOI: 10.1186/s12887-018-1082-2
Development and validation of a diagnostic model for early differentiation of sepsis and non-infectious SIRS in critically ill children - a data-driven approach using machine-learning algorithms
Abstract
Background: Since early antimicrobial therapy is mandatory in septic patients, immediate diagnosis and distinction from non-infectious SIRS is essential but hampered by the similarity of symptoms between both entities. We aimed to develop a diagnostic model for differentiation of sepsis and non-infectious SIRS in critically ill children based on routinely available parameters (baseline characteristics, clinical/laboratory parameters, technical/medical support).
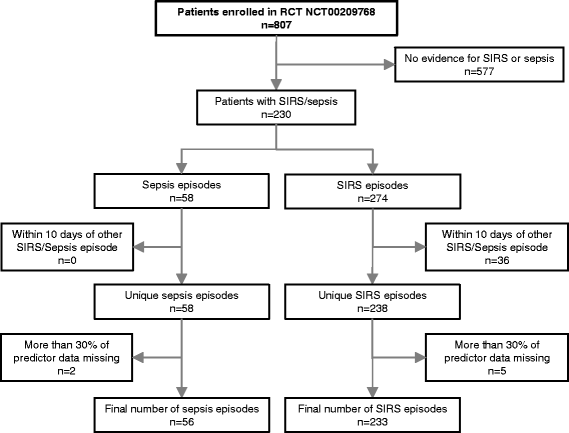
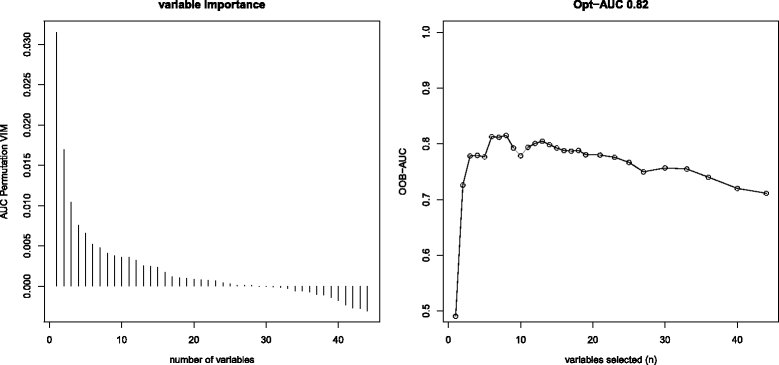
Methods: This is a secondary analysis of a randomized controlled trial conducted at a German tertiary-care pediatric intensive care unit (PICU). Two hundred thirty-eight cases of non-infectious SIRS and 58 cases of sepsis (as defined by IPSCC criteria) were included. We applied a Random Forest approach to identify the best set of predictors out of 44 variables measured at the day of onset of the disease. The developed diagnostic model was validated in a temporal split-sample approach.
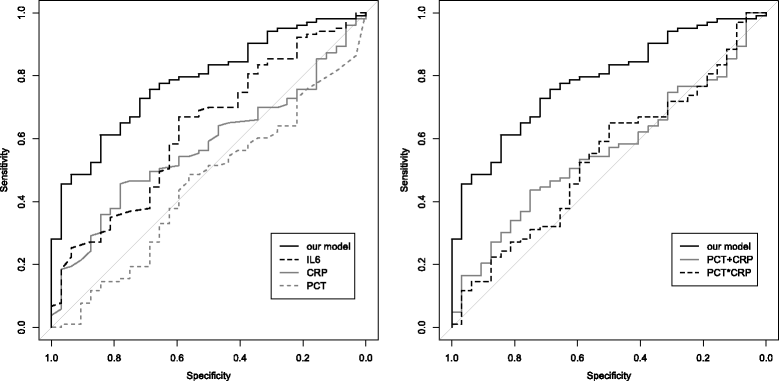
Results: A model including four clinical (length of PICU stay until onset of non-infectious SIRS/sepsis, central line, core temperature, number of non-infectious SIRS/sepsis episodes prior to diagnosis) and four laboratory parameters (interleukin-6, platelet count, procalcitonin, CRP) was identified in the training dataset. Validation in the test dataset revealed an AUC of 0.78 (95% CI: 0.70-0.87). Our model was superior to previously proposed biomarkers such as CRP, interleukin-6, procalcitonin or a combination of CRP and procalcitonin (maximum AUC = 0.63; 95% CI: 0.52-0.74). When aiming at a complete identification of sepsis cases (100%; 95% CI: 87-100%), 28% (95% CI: 20-38%) of non-infectious SIRS cases were assorted correctly.
Conclusions: Our approach allows early recognition of sepsis with an accuracy superior to previously described biomarkers, and could potentially reduce antibiotic use by 30% in non-infectious SIRS cases. External validation studies are necessary to confirm the generalizability of our approach across populations and treatment practices.
Trial registration: ClinicalTrials.gov number: NCT00209768; registration date: September 21, 2005.
Keywords: Diagnosis; Intensive care unit; Pediatric; Random Forest; SIRS; Sepsis.
Conflict of interest statement
Ethics approval and consent to participate
Ethics approval was obtained from the ethics committee of Hannover Medical School (3702/2005). All legal guardians provided written informed consent on admission to PICU.
Consent for publication
Not applicable.
Competing interests
FL, NR, PB, RTM and AK report no conflicts of interest. MS, TJ and MB report having been paid travel and lecture fees from Pall Corporation and B. Braun Corporation.
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Figures
References
-
- Bone RC, Balk RA, Cerra FB, Dellinger RP, Fein AM, Knaus WA et al. American-College of Chest Physicians Society of Critical Care Medicine Consensus Conference - Definitions for Sepsis and Organ Failure and Guidelines for the Use of Innovative Therapies in Sepsis. Crit Care Med. 1992;20:864–74.
-
- Kumar A, Roberts D, Wood KE, Light B, Parrillo JE, Sharma S, Suppes R, Feinstein D, Zanotti S, Taiberg L, et al. Duration of hypotension before initiation of effective antimicrobial therapy is the critical determinant of survival in human septic shock. Crit Care Med. 2006;34:1589–1596. doi: 10.1097/01.CCM.0000217961.75225.E9. - DOI - PubMed
Publication types
MeSH terms
Associated data
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous

